100+ Healthcare and Pharma Project Management SOPs – Excel XLSX
Excel (XLSX)
BENEFITS OF THIS DOWNLOADABLE EXCEL DOCUMENT
- Provides a comprehensive framework for executing healthcare and pharma projects with full lifecycle control, from initiation to close-out.
- Delivers a standardized, audit-ready SOP system aligned with regulatory, clinical, financial, and operational excellence requirements.
- Enables organizations to streamline project execution, reduce risk, and maintain continuous compliance through a structured Excel-based operating model.
HEALTHCARE EXCEL DESCRIPTION
Curated by McKinsey-trained Executives
🚨 100+ HEALTHCARE & PHARMA PROJECT MANAGEMENT SOPs 🚨
🔥 THE ULTIMATE GMP/GCP/GxP-ALIGNED PROJECT OPERATING SYSTEM – BUILT IN EXCEL 🔥
💣 WHY MOST HEALTHCARE & PHARMA PROJECTS FAIL (BRUTAL TRUTH)
Let's cut through the compliance theater:
❌ SOPs scattered across documents no one uses
❌ Regulatory chaos between FDA, EMA, and internal teams
❌ Clinical operations running on tribal knowledge
❌ Risk managed reactively instead of systematically
❌ Budgets leaking due to zero financial governance
❌ Vendor misalignment killing timelines
❌ Audit panic instead of audit readiness
Result?
👉 Delayed clinical trials
👉 Regulatory rejections
👉 Budget overruns
👉 Compliance findings
👉 Data integrity risks
👉 Reputation damage
Let's be honest:
> If your healthcare or pharma projects are not fully SOP-driven, structured, and standardized…
> you don't have control – you have regulated chaos.
🚀 INTRODUCING: THE 150+ HEALTHCARE & PHARMA PROJECT MANAGEMENT SOPs EXCEL LIBRARY
This is not a document pack.
This is not theoretical compliance guidance.
This is a:
✔ 150 End-to-End SOP Execution System
✔ Excel-Based Plug & Play Project Governance Engine
✔ GxP-Ready Operational Framework
✔ Clinical + Regulatory + Financial + Risk Unified System
✔ Audit-Ready, Inspection-Ready, Execution-Ready
📦 WHAT YOU GET
✔ 150 Fully Structured SOPs
✔ 15 Complete Project Management Domains
✔ Excel-Based Instant Deployment
✔ Built-In KPI + Risk + Compliance Layers
✔ Cross-Functional Ownership Framework
✔ End-to-End Lifecycle Coverage (Initiation → Close-Out)
🧠 EVERY SOP IS BUILT FOR EXECUTION (NOT THEORY)
Each SOP includes:
✔ Purpose
✔ Scope
✔ Owner / Role
✔ Inputs (Required Information & Documents)
✔ Process Steps (Detailed Workflow)
✔ Outputs / Deliverables
✔ KPIs / Success Metrics
✔ Risks / Controls
✔ Review Frequency
No fluff. No ambiguity. Just operational control.
📚 FULL 150+ HEALTHCARE & PHARMA SOP LIBRARY
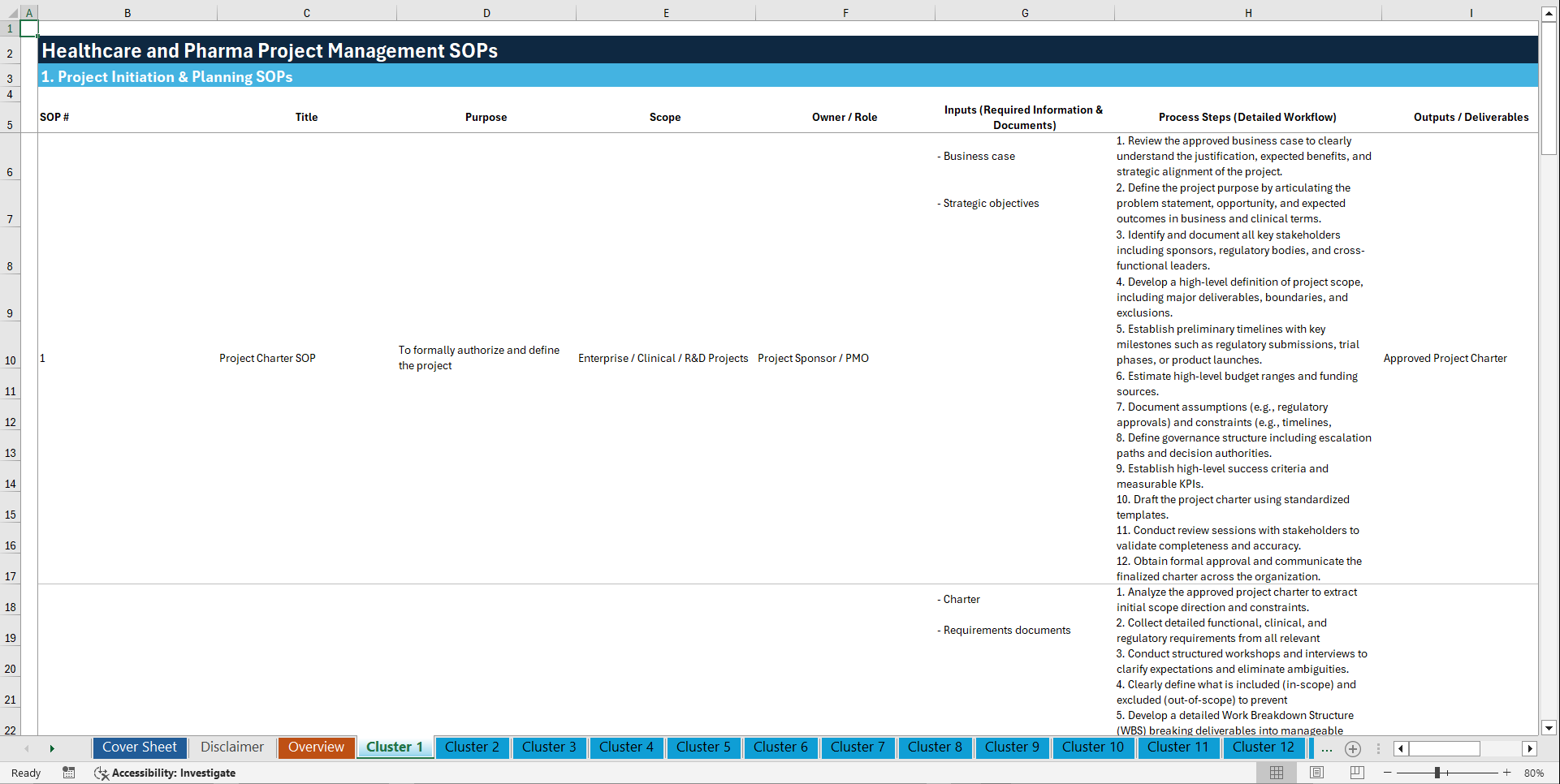
🧩 1. PROJECT INITIATION & PLANNING (1–10)
1. Project Charter SOP
2. Project Scope Definition SOP
3. Project Objectives SOP
4. Stakeholder Identification SOP
5. Project Feasibility Assessment SOP
6. Resource Planning SOP
7. Risk Identification SOP
8. Budget Estimation SOP
9. Project Schedule Development SOP
10. Project Governance SOP
🧩 2. REGULATORY COMPLIANCE (11–20)
11. Regulatory Requirements Assessment SOP
12. FDA Submission SOP
13. EMA Submission SOP
14. Health Authority Communication SOP
15. Clinical Trial Approval SOP
16. Regulatory Documentation SOP
17. Compliance Monitoring SOP
18. Change Control SOP
19. CAPA Management SOP
20. Audit Readiness SOP
🧩 3. CLINICAL PROJECT MANAGEMENT (21–30)
21. Clinical Trial Planning SOP
22. Protocol Development SOP
23. Investigator Selection SOP
24. Site Initiation SOP
25. Subject Recruitment SOP
26. Informed Consent SOP
27. Study Monitoring SOP
28. Data Collection SOP
29. Adverse Event Reporting SOP
30. Clinical Study Close-Out SOP
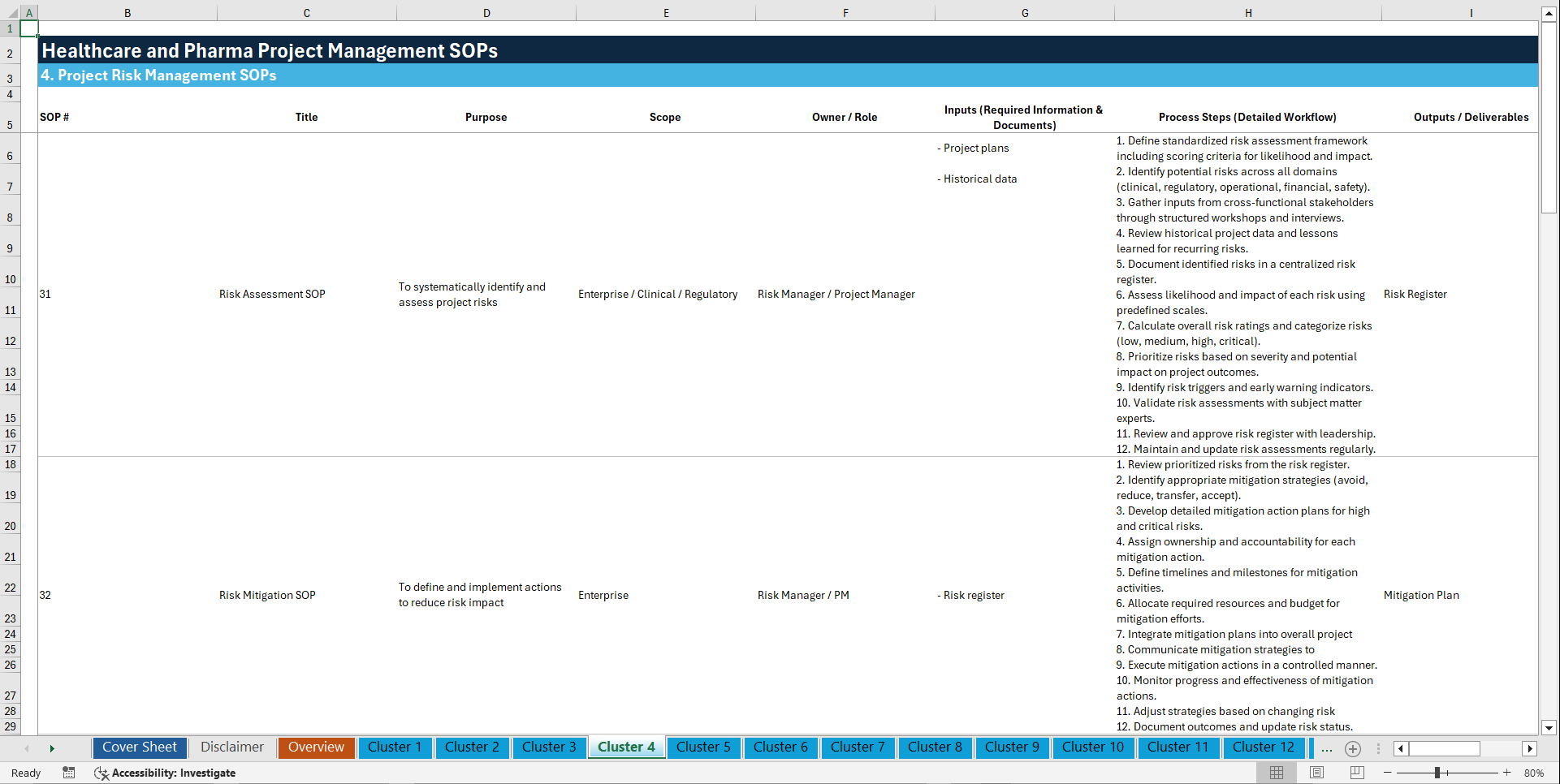
🧩 4. PROJECT RISK MANAGEMENT (31–40)
31. Risk Assessment SOP
32. Risk Mitigation SOP
33. Risk Escalation SOP
34. Contingency Planning SOP
35. Quality Risk Management SOP
36. Risk Communication SOP
37. Vendor Risk Assessment SOP
38. Change Risk Evaluation SOP
39. Health & Safety Risk SOP
40. Project Risk Review SOP
🧩 5. BUDGET & FINANCIAL MANAGEMENT (41–50)
41. Budget Planning SOP
42. Cost Tracking SOP
43. Financial Reporting SOP
44. Resource Cost Allocation SOP
45. Contract Budgeting SOP
46. Procurement SOP
47. Vendor Payment SOP
48. Expense Approval SOP
49. Financial Risk Assessment SOP
50. Budget Variance Management SOP
🧩 6. RESOURCE & TEAM MANAGEMENT (51–60)
51. Project Team Formation SOP
52. Roles & Responsibilities SOP
53. Resource Allocation SOP
54. Staff Onboarding SOP
55. Training Plan SOP
56. Performance Monitoring SOP
57. Conflict Resolution SOP
58. Team Communication SOP
59. Knowledge Transfer SOP
60. Staff Offboarding SOP
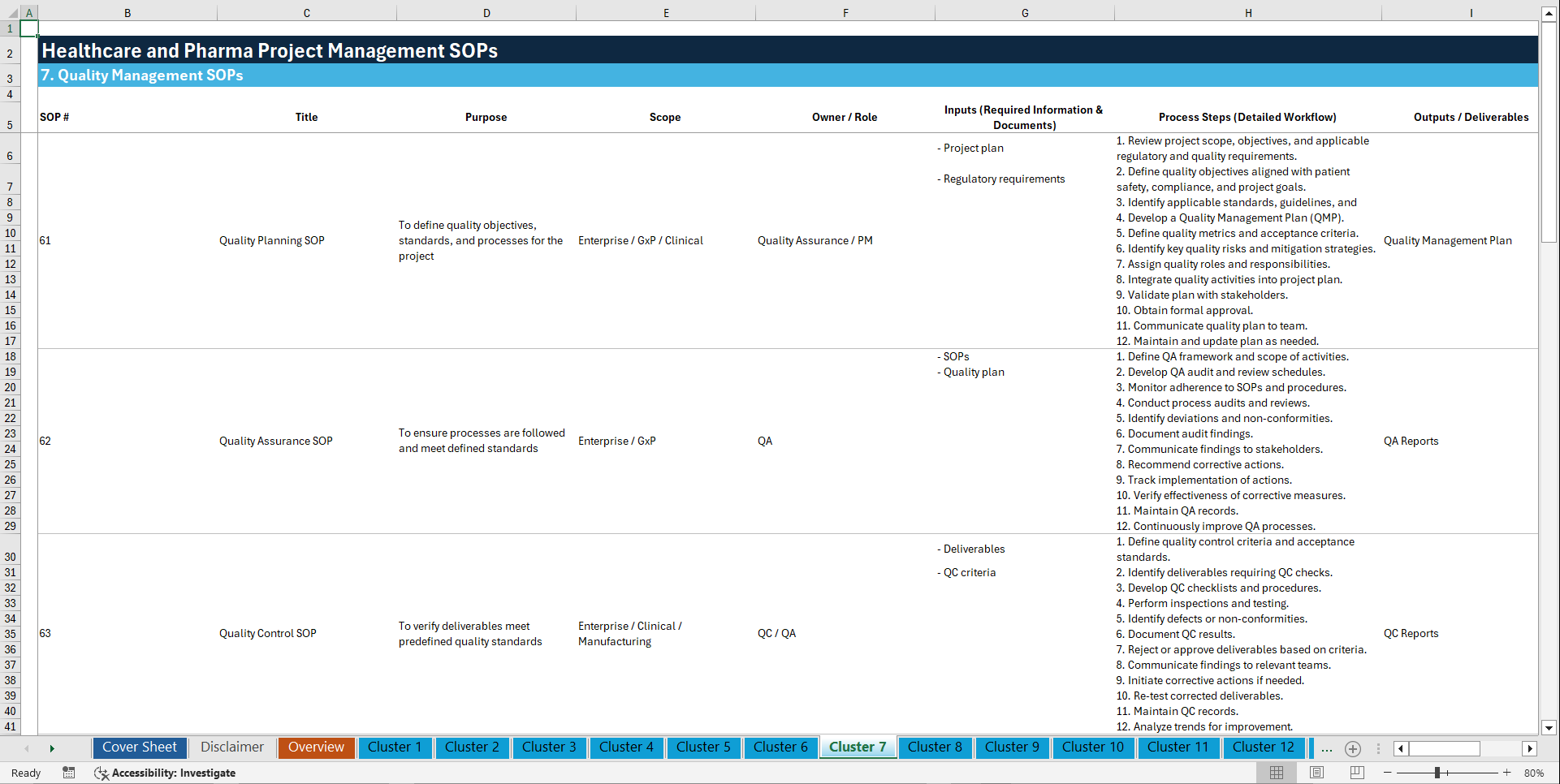
🧩 7. QUALITY MANAGEMENT (61–70)
61. Quality Planning SOP
62. Quality Assurance SOP
63. Quality Control SOP
64. Process Validation SOP
65. SOP Review & Approval SOP
66. Document Control SOP
67. Audit Management SOP
68. Corrective Action SOP
69. Preventive Action SOP
70. Continuous Improvement SOP
🧩 8. VENDOR & CONTRACT MANAGEMENT (71–80)
71. Vendor Selection SOP
72. Vendor Qualification SOP
73. Contract Negotiation SOP
74. Contract Review SOP
75. Vendor Performance Monitoring SOP
76. Service Level Agreement SOP
77. Purchase Order SOP
78. Vendor Audit SOP
79. Subcontractor Management SOP
80. Termination & Transition SOP
🧩 9. PROJECT MONITORING & REPORTING (81–90)
81. Project Status Reporting SOP
82. KPI Tracking SOP
83. Milestone Tracking SOP
84. Progress Review SOP
85. Dashboard Management SOP
86. Issue Logging SOP
87. Change Request SOP
88. Escalation SOP
89. Performance Metrics SOP
90. Lessons Learned SOP
🧩 10. DOCUMENT & DATA MANAGEMENT (91–100)
91. Document Creation SOP
92. Document Review SOP
93. Document Approval SOP
94. Document Archiving SOP
95. Data Management SOP
96. Data Entry SOP
97. Data Validation SOP
98. Data Security SOP
99. Electronic Records SOP
100. Data Backup & Recovery SOP
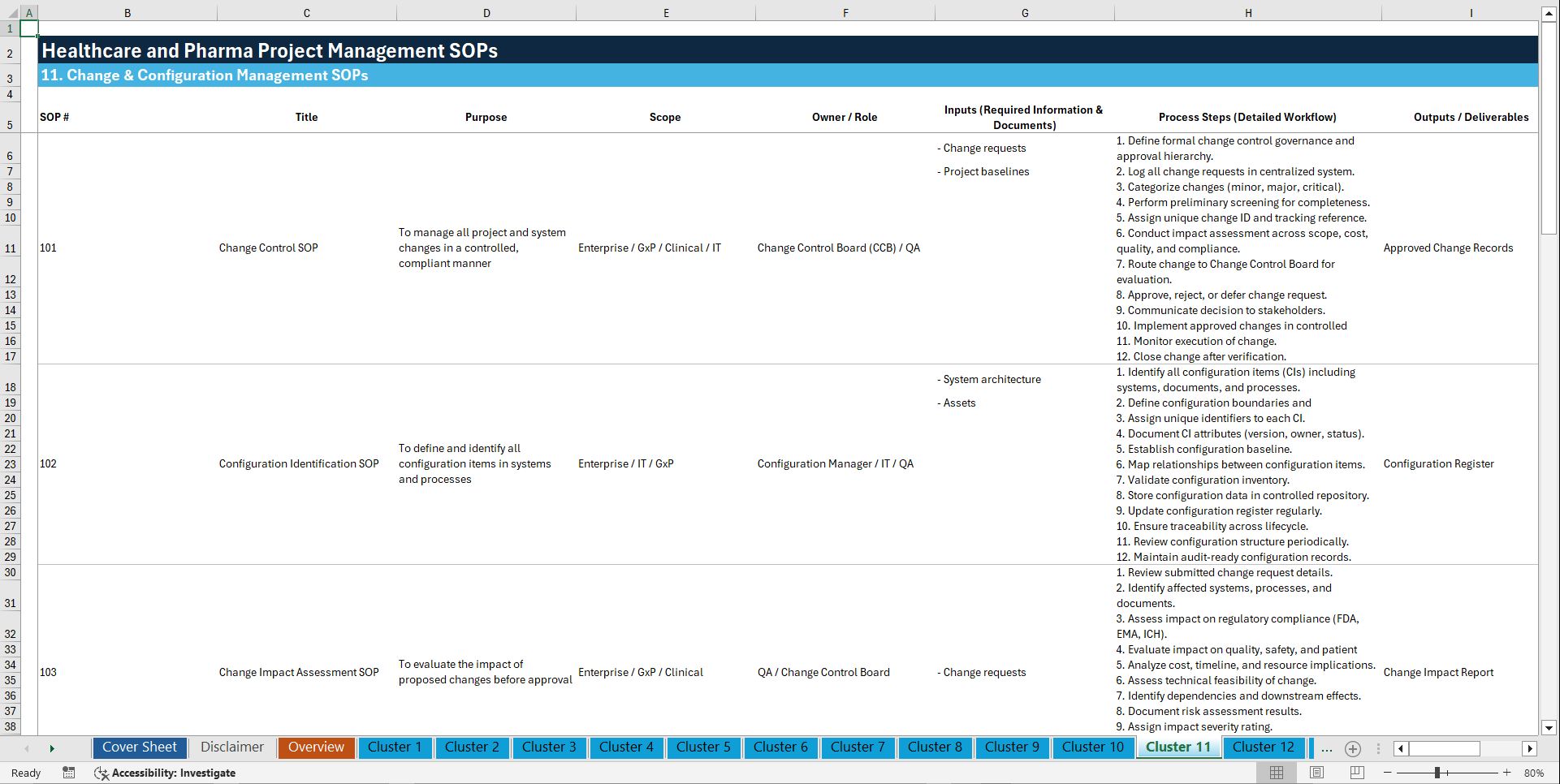
🧩 11. CHANGE & CONFIGURATION MANAGEMENT (101–110)
101. Change Control SOP
102. Configuration Identification SOP
103. Change Impact Assessment SOP
104. Change Implementation SOP
105. Change Communication SOP
106. Version Control SOP
107. Deviation Management SOP
108. Engineering Change Request SOP
109. Document Change SOP
110. Change Closure SOP
🧩 12. HEALTH, SAFETY & ENVIRONMENT (111–120)
111. Workplace Safety SOP
112. Hazard Identification SOP
113. Emergency Response SOP
114. Incident Reporting SOP
115. Safety Training SOP
116. PPE Management SOP
117. Waste Management SOP
118. Environmental Compliance SOP
119. Safety Audits SOP
120. Risk Assessment for HSE SOP
🧩 13. TECHNOLOGY & IT MANAGEMENT (121–130)
121. Project Management Software SOP
122. IT Infrastructure SOP
123. System Validation SOP
124. User Access Management SOP
125. Cybersecurity SOP
126. Data Integrity SOP
127. Electronic Signature SOP
128. System Backup SOP
129. IT Change Management SOP
130. IT Incident Management SOP
🧩 14. CLINICAL SUPPLY & LOGISTICS (131–140)
131. Supply Chain Planning SOP
132. Material Procurement SOP
133. Inventory Management SOP
134. Storage & Handling SOP
135. Temperature Monitoring SOP
136. Shipment SOP
137. Distribution SOP
138. Returns & Disposal SOP
139. Labeling SOP
140. Supply Chain Risk Management SOP
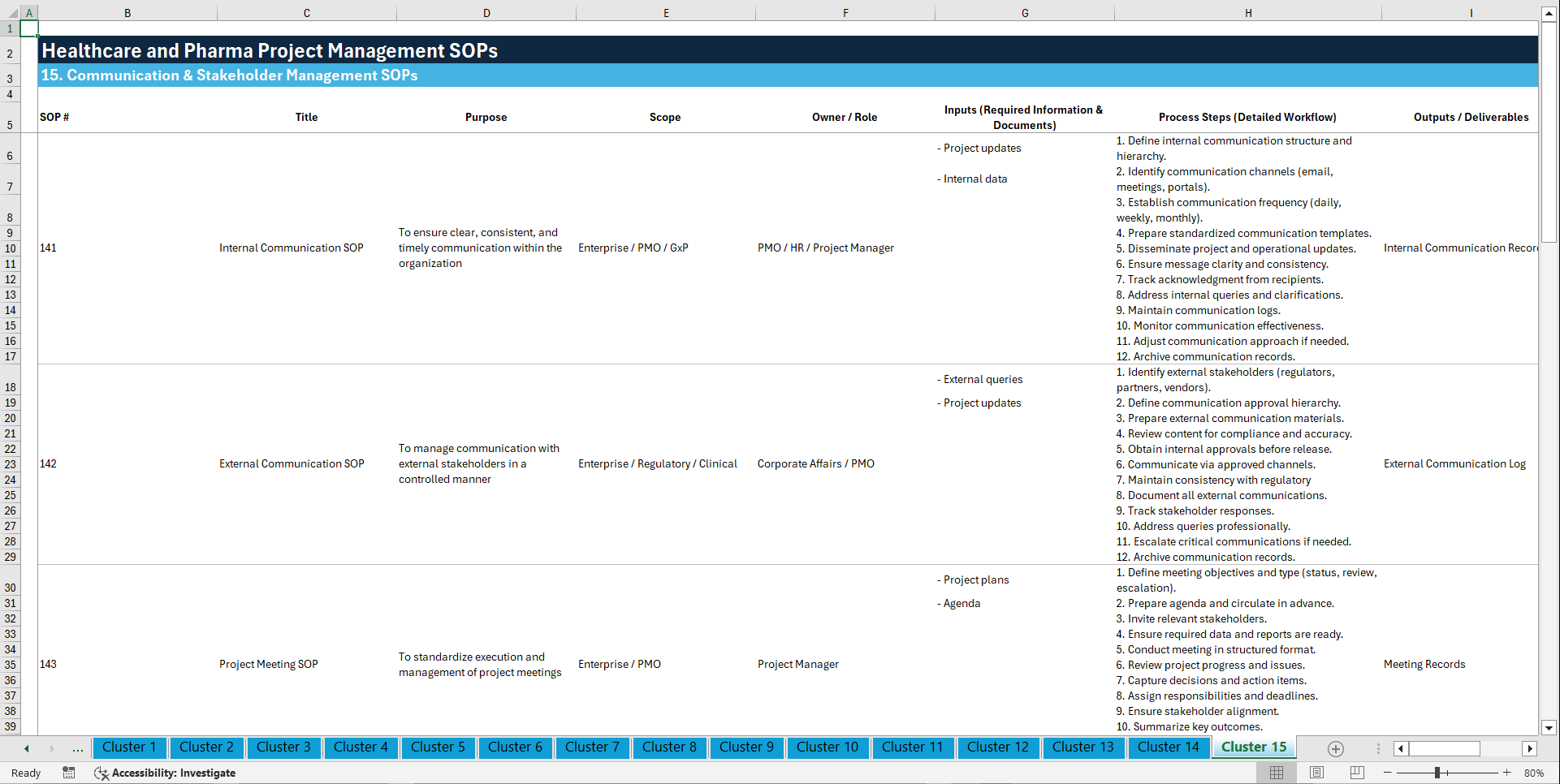
🧩 15. COMMUNICATION & STAKEHOLDER MANAGEMENT (141–150)
141. Internal Communication SOP
142. External Communication SOP
143. Project Meeting SOP
144. Stakeholder Reporting SOP
145. Meeting Minutes SOP
146. Communication Plan SOP
147. Feedback Management SOP
148. Escalation SOP
149. Public Relations SOP
150. Knowledge Sharing SOP
🎯 WHAT THIS SYSTEM DOES FOR YOU
🚀 Transforms fragmented operations into a unified execution engine
📊 Standardizes ALL healthcare & pharma project workflows
🔒 Ensures regulatory and audit readiness at all times
⚡ Accelerates clinical and operational timelines
💰 Eliminates financial leakage and budget inefficiencies
📉 Reduces compliance, operational, and project risks
🧠 Creates true enterprise-level process maturity
⚡ FINAL TRUTH
Healthcare & pharma leaders don't win because they "work harder."
They win because:
✔ They systemize execution
✔ They standardize processes
✔ They control risk proactively
✔ They operationalize compliance
Meanwhile everyone else:
❌ Reacts
❌ Scrambles
❌ Fails audits
❌ Misses timelines
🚀 THIS IS NOT JUST AN SOP LIBRARY.
💣 THIS IS YOUR HEALTHCARE & PHARMA PROJECT OPERATING SYSTEM.
👉 IF YOU WANT CONTROL, COMPLIANCE, AND SCALABILITY – YOU START WITH SYSTEMS.
Key Words:
Strategy & Transformation, Growth Strategy, Strategic Planning, Strategy Frameworks, Innovation Management, Pricing Strategy, Core Competencies, Strategy Development, Business Transformation, Marketing Plan Development, Product Strategy, Breakout Strategy, Competitive Advantage, Mission, Vision, Values, Strategy Deployment & Execution, Innovation, Vision Statement, Core Competencies Analysis, Corporate Strategy, Product Launch Strategy, BMI, Blue Ocean Strategy, Breakthrough Strategy, Business Model Innovation, Business Strategy Example, Corporate Transformation, Critical Success Factors, Customer Segmentation, Customer Value Proposition, Distinctive Capabilities, Enterprise Performance Management, KPI, Key Performance Indicators, Market Analysis, Market Entry Example, Market Entry Plan, Market Intelligence, Market Research, Market Segmentation, Market Sizing, Marketing, Michael Porter's Value Chain, Organizational Transformation, Performance Management, Performance Measurement, Platform Strategy, Product Go-to-Market Strategy, Reorganization, Restructuring, SWOT, SWOT Analysis, Service 4.0, Service Strategy, Service Transformation, Strategic Analysis, Strategic Plan Example, Strategy Deployment, Strategy Execution, Strategy Frameworks Compilation, Strategy Methodologies, Strategy Report Example, Value Chain, Value Chain Analysis, Value Innovation, Value Proposition, Vision Statement, Corporate Strategy, Business Development, Business plan pdf, business plan, PDF, Business Plan DOC, Business Plan Template, PPT, Market strategy playbook, strategic market planning, competitive analysis tools, market segmentation frameworks, growth strategy templates, product positioning strategy, market execution toolkit, strategic alignment playbook, KPI and OKR frameworks, business growth strategy guide, cross-functional strategy templates, market risk management, market strategy PowerPoint doc, guide, ebook, e-book ,McKinsey Change Playbook, Organizational change management toolkit, Change management frameworks 2025, Influence model for change, Change leadership strategies, Behavioral change in organizations, Change management PowerPoint templates, Transformational leadership in change, supply chain KPIs, supply chain KPI toolkit, supply chain PowerPoint template, logistics KPIs, procurement KPIs, inventory management KPIs, supply chain performance metrics, manufacturing KPIs, supply chain dashboard, supply chain strategy KPIs, reverse logistics KPIs, sustainability KPIs in supply chain, financial supply chain KPIs, warehouse KPIs, digital supply chain KPIs, 1200 KPIs, supply chain scorecard, KPI examples, supply chain templates, Corporate Finance SOPs, Finance SOP Excel Template, CFO Toolkit, Finance Department Procedures, Financial Planning SOPs, Treasury SOPs, Accounts Payable SOPs, Accounts Receivable SOPs, General Ledger SOPs, Accounting Policies Template, Internal Controls SOPs, Finance Process Standardization, Finance Operating Procedures, Finance Department Excel Template, FP&A Process Documentation, Corporate Finance Template, Finance SOP Toolkit, CFO Process Templates, Accounting SOP Package, Tax Compliance SOPs, Financial Risk Management Procedures.
NOTE: Our digital products are sold on an "as is" basis, making returns and refunds unavailable post-download. Please preview and inquire before purchasing. Please contact us before purchasing if you have any questions! This policy aligns with the standard Flevy Terms of Usage.
Got a question about the product? Email us at support@flevy.com or ask the author directly by using the "Ask the Author a Question" form. If you cannot view the preview above this document description, go here to view the large preview instead.
Source: Best Practices in Healthcare, Pharma Excel: 100+ Healthcare and Pharma Project Management SOPs Excel (XLSX) Spreadsheet, SB Consulting