Generative AI (GenAI) in the Pharmaceutical Industry – PowerPoint PPTX Template
PowerPoint (PPTX) 33 Slides FlevyPro Document
ARTIFICIAL INTELLIGENCE PPT TEMPLATE DESCRIPTION
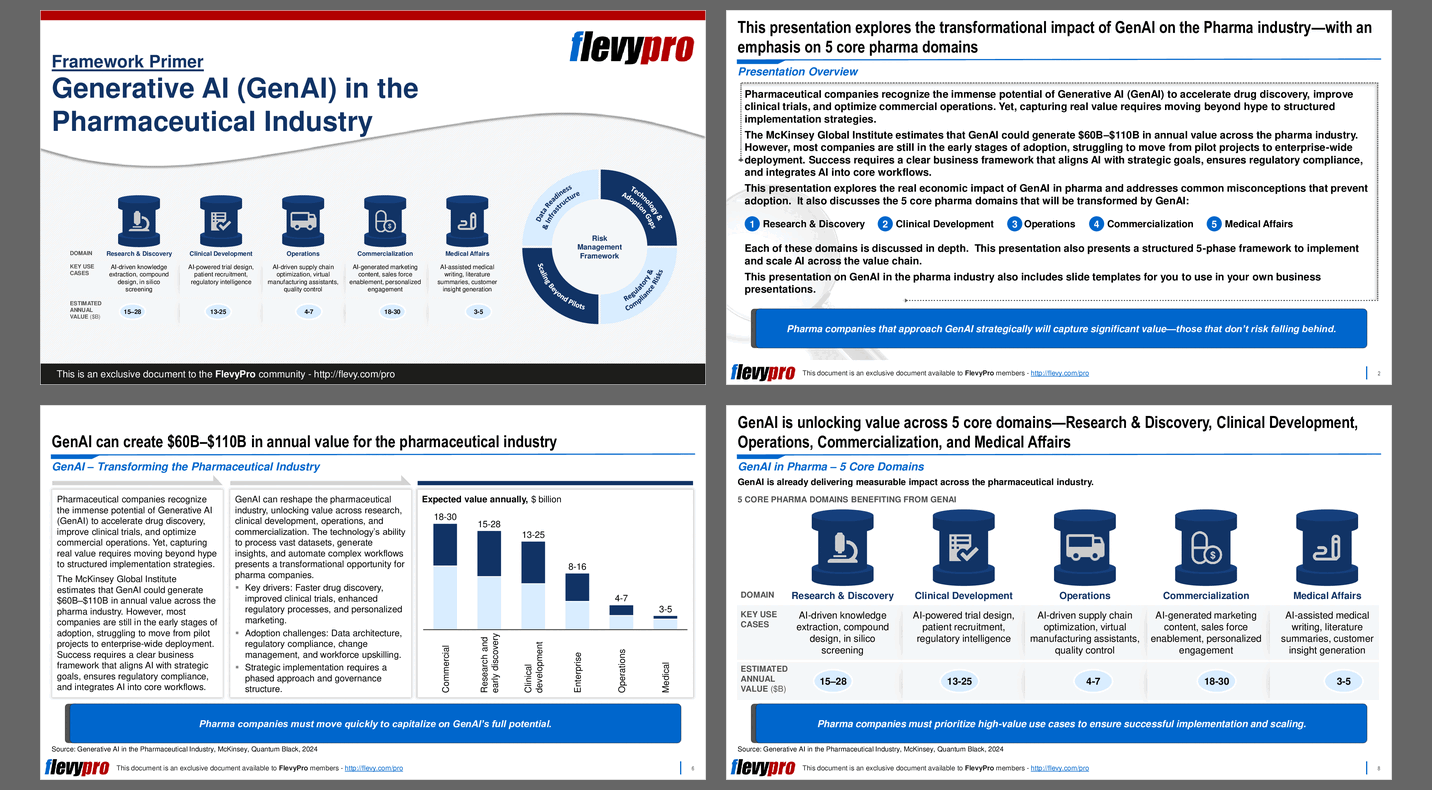
Pharmaceutical companies recognize the immense potential of Generative AI (GenAI) to accelerate drug discovery, improve clinical trials, and optimize commercial operations. Yet, capturing real value requires moving beyond hype to structured implementation strategies.
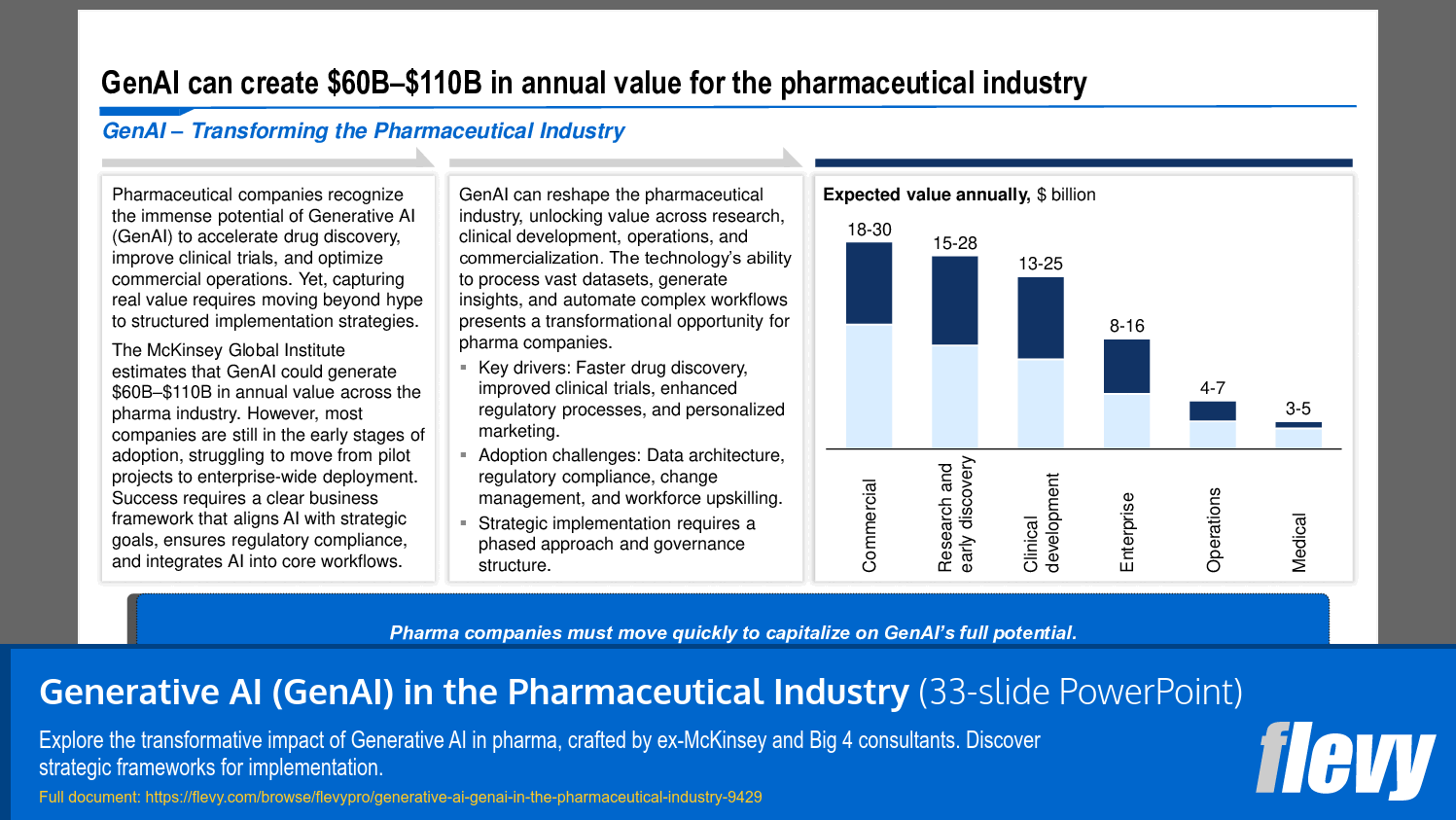
The McKinsey Global Institute estimates that GenAI could generate $60B–$110B in annual value across the pharma industry. However, most companies are still in the early stages of adoption, struggling to move from pilot projects to enterprise-wide deployment. Success requires a clear business framework that aligns AI with strategic goals, ensures regulatory compliance, and integrates AI into core workflows.
This PPT presentation explores the real economic impact of GenAI in pharma and addresses common misconceptions that prevent adoption. It also discusses the 5 core pharma domains that will be transformed by GenAI:
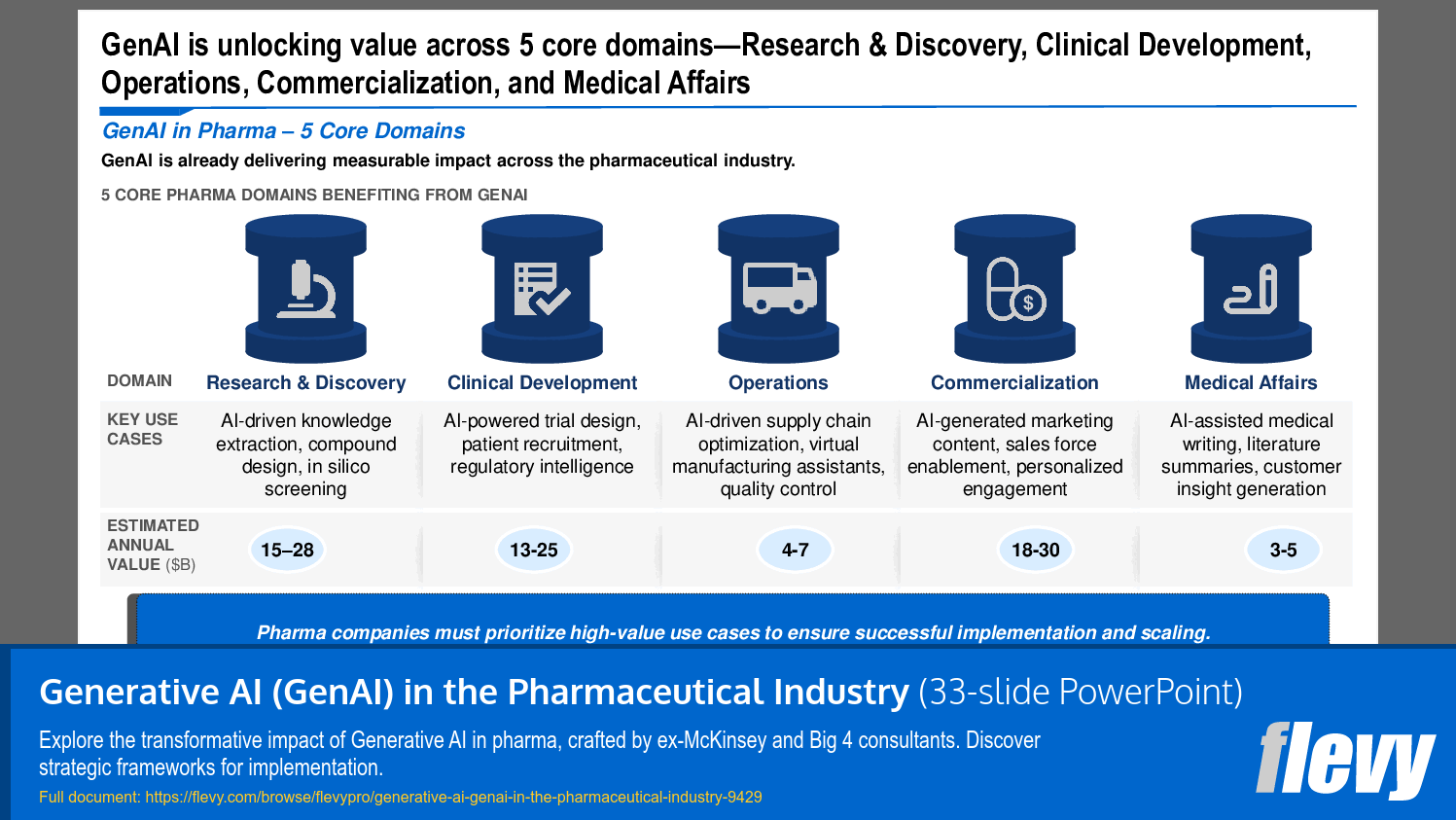
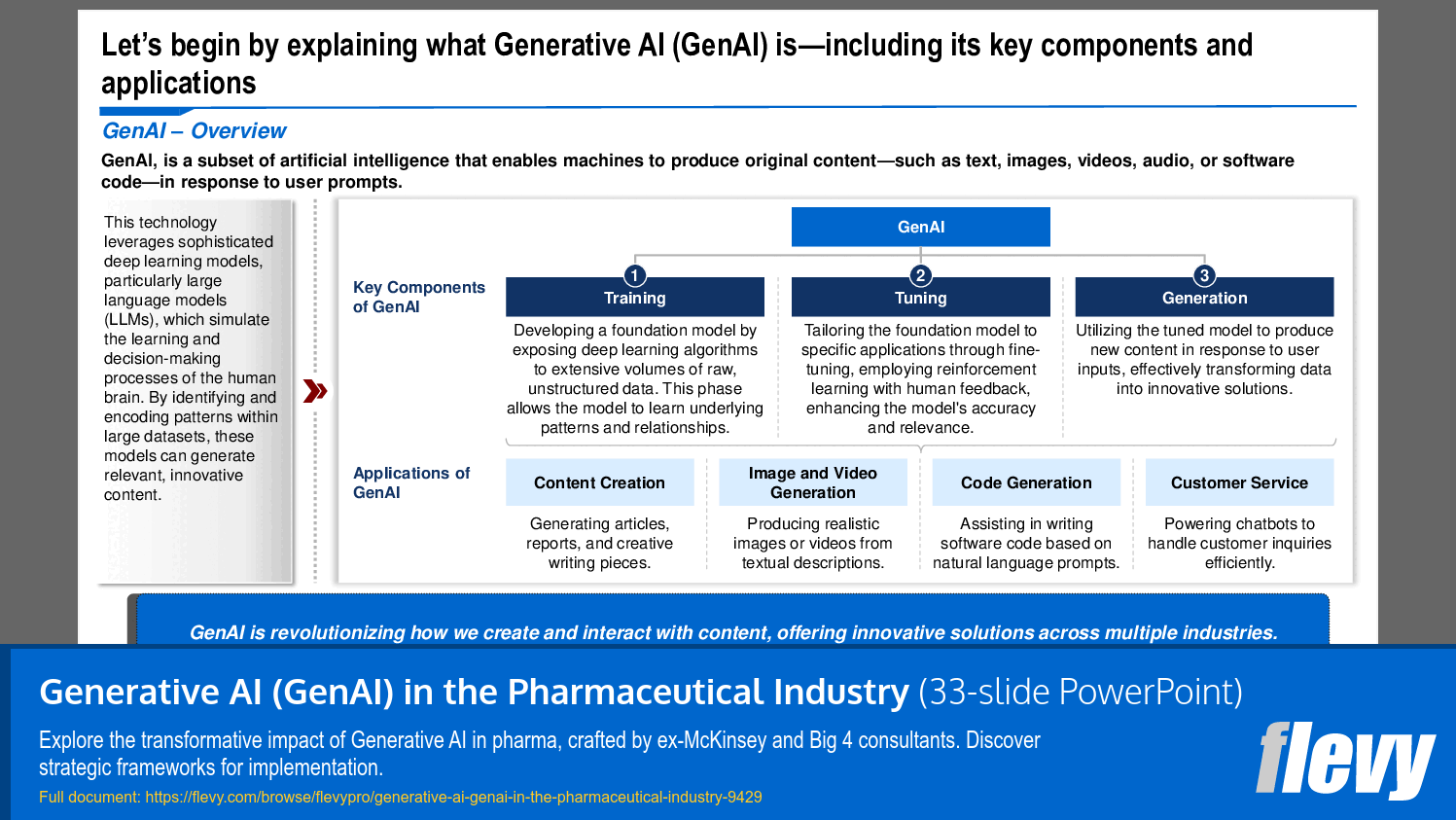
1. Research & Discovery – Utilize GenAI to drive the early-stage identification and development of new pharmaceutical compounds through AI-powered knowledge extraction, compound design, and in silico screening, which involves virtual simulations to predict molecular interactions.
2. Clinical Development – Deploy GenAI solutions to streamline the design, execution, and management of clinical trials by leveraging AI for trial design, patient recruitment, regulatory intelligence, and the optimization of virtual trials.
3. Operations – Use GenAI to drive improvements around the efficiency and reliability of pharmaceutical production and logistics through AI-driven supply chain optimization, virtual manufacturing assistance, and quality control processes.
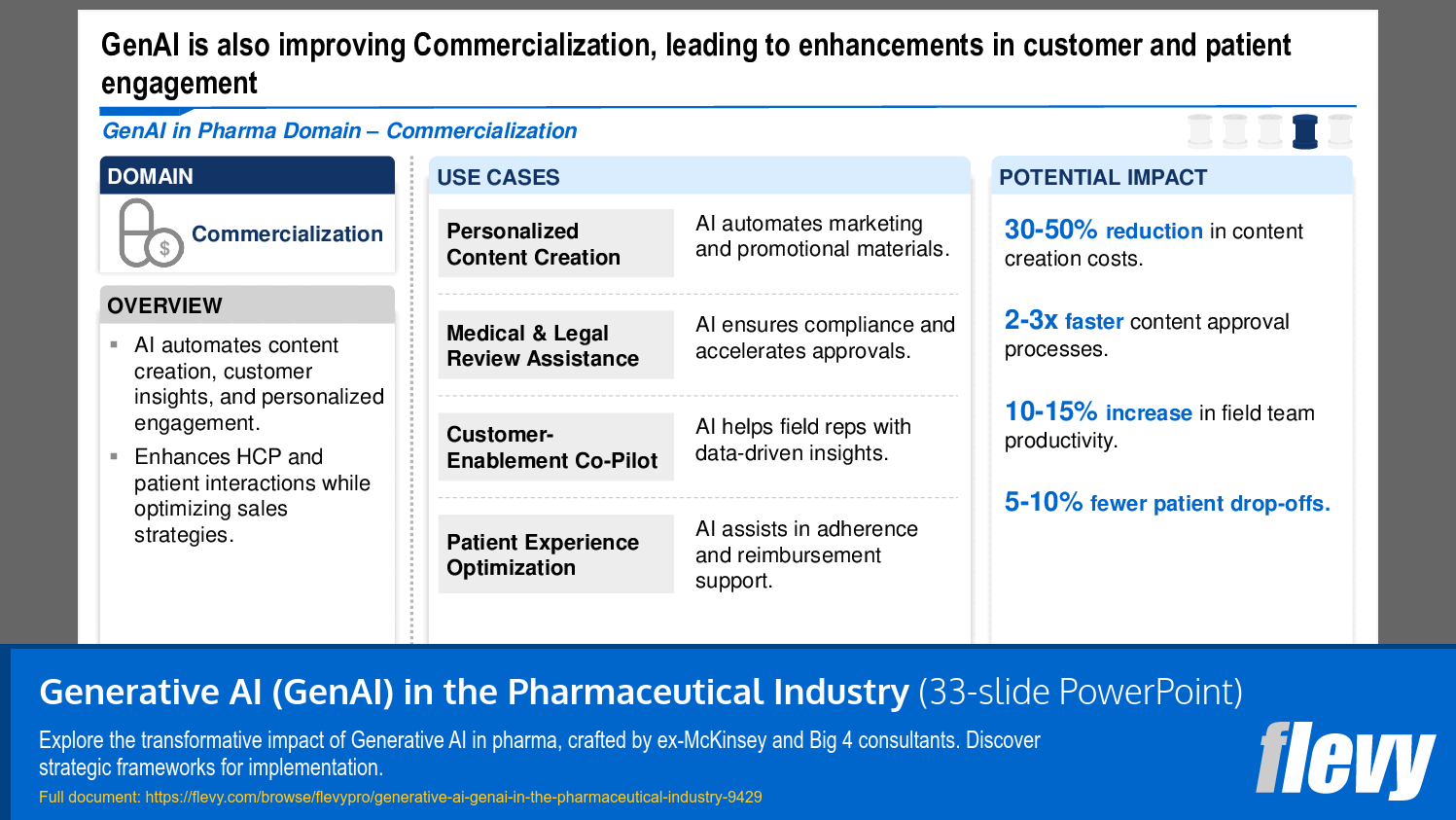
4. Commercialization – Improve the marketing and sales of pharmaceutical products by generating AI-driven marketing content, enabling sales force effectiveness, and providing personalized customer engagement strategies.
5. Medical Affairs – Support post-market activities and medical communication by facilitating AI-assisted medical writing, generating literature summaries, and producing customer insights for better stakeholder engagement.
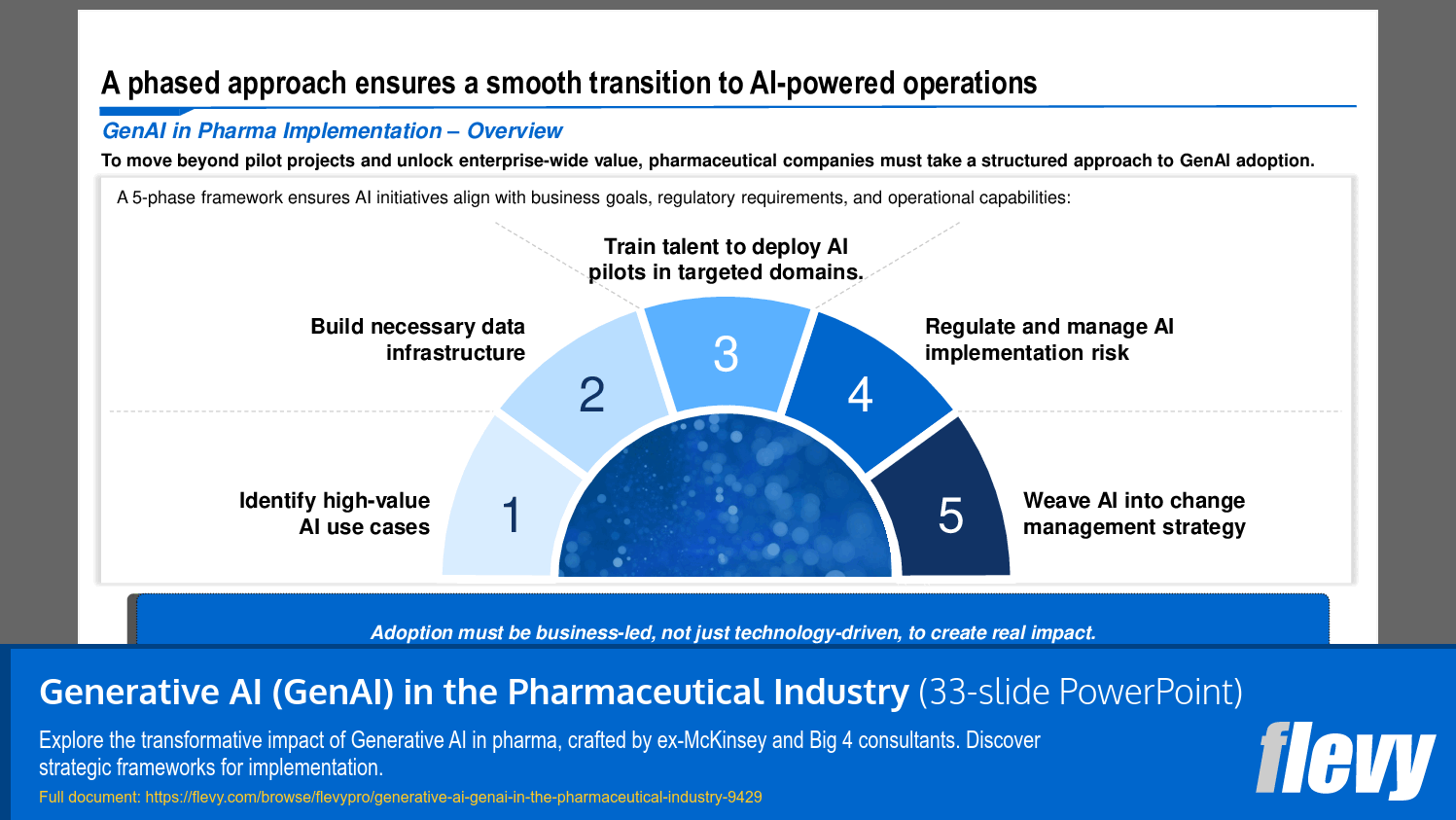
Each of these domains is discussed in depth. This presentation also presents a structured 5-phase framework to implement and scale AI across the value chain.
This PPT presentation on GenAI in the pharma industry also includes slide templates for you to use in your own business presentations.
Got a question about this document? Email us at flevypro@flevy.com.
PRESENTATION DEEP DIVE ANALYSIS
This deep-dive analysis was generated from the full 33-slide PowerPoint presentation.
Executive Summary
The "Generative AI (GenAI) in the Pharmaceutical Industry" presentation provides a comprehensive exploration of how GenAI can revolutionize various aspects of the pharmaceutical sector. It highlights 5 core domains—Research & Discovery, Clinical Development, Operations, Commercialization, and Medical Affairs—where GenAI can drive significant value. With the potential to generate $60B–$110B annually, the presentation outlines structured frameworks for effective implementation, addressing common misconceptions and emphasizing the importance of aligning AI initiatives with strategic business goals.
Who This Is For and When to Use
• Pharmaceutical executives seeking to leverage AI for operational improvements
• Research and development teams focused on accelerating drug discovery
• Clinical trial managers aiming to enhance trial efficiency and patient recruitment
• Marketing and sales professionals looking to optimize customer engagement
• Compliance officers ensuring adherence to regulatory standards
Best-fit moments to use this deck:
• Strategic planning sessions for AI integration in pharmaceutical operations
• Workshops on improving clinical trial processes through technology
• Executive briefings on the economic impact of AI in the pharma sector
• Training sessions for teams on implementing AI-driven solutions
Learning Objectives
• Define the role of Generative AI in transforming pharmaceutical operations
• Identify high-value AI use cases across the pharmaceutical value chain
• Build necessary data infrastructure to support AI initiatives
• Train talent to effectively deploy AI pilots in targeted domains
• Regulate and manage AI implementation risks to ensure compliance
• Weave AI into change management strategies for seamless integration
Table of Contents
• Overview (page 1)
• GenAI (page 4)
• GenAI in Pharma (page 8)
• GenAI in Pharma Implementation (page 15)
• Slide Design Structure & Templates (page 20)
Primary Topics Covered
• Generative AI Overview - GenAI is a subset of AI that enables machines to produce original content, transforming industries by enhancing creativity and efficiency.
• Core Domains of GenAI in Pharma - The presentation identifies 5 domains where GenAI can create substantial value, including Research & Discovery and Clinical Development.
• Implementation Framework - A structured five-phase framework guides organizations in adopting AI, ensuring alignment with business objectives and regulatory compliance.
• Economic Impact of GenAI - The McKinsey Global Institute estimates that GenAI could unlock $60B–$110B in annual value for the pharmaceutical industry.
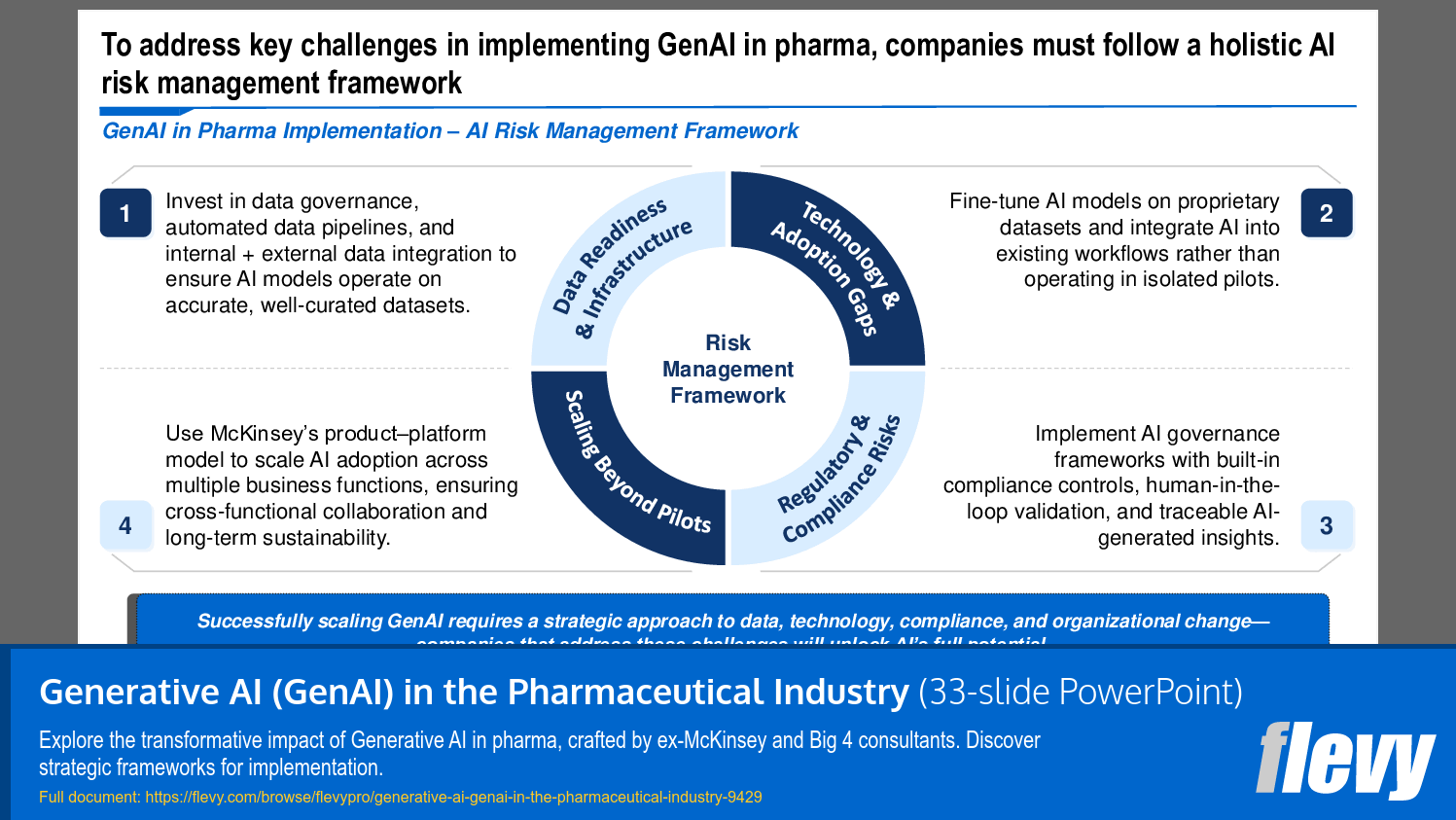
• Challenges in AI Adoption - Common barriers include data readiness, technology gaps, regulatory compliance, and scaling beyond pilot projects.
• AI Risk Management Framework - A holistic approach to managing risks associated with AI implementation in the pharmaceutical sector, focusing on data integrity and compliance.
Deliverables, Templates, and Tools
• Framework for identifying high-value AI use cases
• Data infrastructure assessment template for AI readiness
• Training module for deploying AI pilots in pharma
• Risk management checklist for AI implementation
• Change management strategy template for integrating AI solutions
• Slide templates for presenting GenAI initiatives
Slide Highlights
• Overview of the economic potential of GenAI in the pharmaceutical industry
• Detailed breakdown of the 5 core domains benefiting from GenAI
• Visual representation of the implementation framework and phases
• Key challenges and solutions for successful AI adoption in pharma
• AI risk management framework outlining critical focus areas
Potential Workshop Agenda
Identifying AI Use Cases Workshop (90 minutes)
• Discuss high-value AI applications in pharmaceutical operations
• Brainstorm potential pilot projects and their expected outcomes
• Assign teams to develop initial project proposals
Data Infrastructure Planning Session (60 minutes)
• Review current data readiness and identify gaps
• Outline steps for building necessary data infrastructure
• Establish timelines for data integration efforts
AI Implementation Strategy Meeting (120 minutes)
• Align on the five-phase framework for AI deployment
• Define roles and responsibilities for each phase
• Develop a roadmap for training and change management
Customization Guidance
• Tailor the data infrastructure assessment to reflect specific organizational needs
• Adjust the implementation framework to align with existing business processes
• Incorporate company-specific terminology and metrics into the templates provided
• Modify the risk management framework to address unique regulatory requirements
Secondary Topics Covered
• AI-driven knowledge extraction and compound design
• AI-powered trial design and patient recruitment strategies
• AI applications in supply chain optimization and quality control
• Enhancements in marketing content creation and customer engagement
• AI-assisted medical writing and literature summarization
Topic FAQ
What are the main areas where Generative AI can add value in the pharmaceutical industry?
GenAI can create value across 5 core domains: Research & Discovery, Clinical Development, Operations, Commercialization, and Medical Affairs. Each domain includes specific applications such as compound design, trial optimization, supply-chain improvements, marketing content generation, and medical writing, organized across the presentation’s 5 core domains.What does a five-phase implementation framework for GenAI typically emphasize?
A five-phase implementation framework for GenAI emphasizes aligning AI initiatives with strategic business goals, assessing data readiness, addressing technology gaps, managing regulatory compliance, and planning for scaling beyond pilots. The presentation structures adoption around these stages and presents a visual five-phase framework.What data infrastructure components are necessary to support GenAI initiatives in pharma?
Supporting GenAI requires high-quality, structured data and systems for collection, storage, and analysis to enable model training and inference; organizations should assess gaps in data readiness and integration. The deck includes a data infrastructure assessment template to guide those evaluations.How can GenAI be applied to improve clinical trial processes?
GenAI can support trial design, optimize patient recruitment, provide regulatory intelligence, and enable virtual trial optimization to increase efficiency and reduce timelines. The presentation details clinical development use cases such as trial design improvements and patient recruitment strategies.What should buyers look for when choosing a GenAI strategy deck or toolkit for pharma?
Buyers should seek coverage of the 5 core pharma domains, a clear implementation framework, templates for data readiness, risk and change management, and presentation-ready slides for executive briefings. The Flevy product provides these elements, including data infrastructure and risk management templates.How should organizations prioritize GenAI use cases for pilot projects?
Prioritization should assess alignment with strategic goals, expected ROI, feasibility given current data and technology, and regulatory constraints. The presentation supplies a framework for identifying high-value AI use cases to guide pilot selection and sequencing.What are common barriers to scaling GenAI pilots across a pharmaceutical company?
Common barriers include insufficient data readiness, technology integration gaps, regulatory compliance challenges, and difficulties scaling beyond pilot projects into enterprise workflows. The deck highlights these adoption challenges and approaches to overcome them, such as addressing data readiness and governance.What risk management practices are important when deploying GenAI in pharma?
Risk management should focus on ensuring data integrity, maintaining regulatory compliance, closing technology gaps, and establishing governance for scaling and monitoring models. The presentation includes a risk management checklist to operationalize these practices.Document FAQ
These are questions addressed within this presentation.
What is Generative AI (GenAI)?
GenAI is a subset of artificial intelligence that enables machines to create original content, including text, images, and more, based on user prompts.
How can GenAI benefit the pharmaceutical industry?
GenAI can enhance drug discovery, streamline clinical trials, optimize operations, and improve customer engagement, potentially generating significant economic value.
What are the core domains where GenAI is applied in pharma?
The 5 core domains are Research & Discovery, Clinical Development, Operations, Commercialization, and Medical Affairs.
What challenges do companies face in adopting GenAI?
Key challenges include data readiness, technology integration, regulatory compliance, and the ability to scale beyond pilot projects.
How can companies ensure successful AI implementation?
A structured approach that aligns AI initiatives with business goals, invests in data infrastructure, and emphasizes change management is essential for success.
What is the estimated annual value that GenAI can generate for pharma?
The McKinsey Global Institute estimates that GenAI could create $60B–$110B in annual value across the pharmaceutical industry.
How does the AI risk management framework work?
The framework focuses on ensuring data readiness, addressing technology gaps, maintaining regulatory compliance, and facilitating scaling beyond initial pilots.
What training is necessary for deploying AI in pharma?
Training should focus on upskilling teams to effectively use AI tools, understanding regulatory requirements, and fostering collaboration among cross-functional teams.
How can organizations prioritize AI use cases?
Organizations should assess potential use cases based on their alignment with strategic goals, expected ROI, and feasibility of implementation.
What templates are included in the presentation?
Templates include frameworks for identifying AI use cases, data infrastructure assessments, training modules, and risk management checklists.
Glossary
• Generative AI (GenAI) - A subset of AI that enables machines to create original content.
• Clinical Development - The phase in drug development focused on testing and evaluating new drugs in humans.
• Data Infrastructure - The systems and processes that support data collection, storage, and analysis.
• Risk Management Framework - A structured approach to identifying, assessing, and mitigating risks associated with AI implementation.
• Change Management - The process of managing the transition of individuals, teams, and organizations to a desired future state.
• AI Use Cases - Specific applications of AI technology to solve business problems or enhance operations.
• Regulatory Compliance - Adherence to laws, regulations, guidelines, and specifications relevant to business processes.
• Training Module - A structured program designed to educate employees on specific skills or knowledge areas.
• Implementation Framework - A structured approach to deploying new technologies or processes within an organization.
• Data Readiness - The state of having high-quality, structured data available for analysis and decision-making.
• Technology Adoption Gaps - Discrepancies between the current technology capabilities and the desired technology outcomes.
• Scaling Beyond Pilots - The process of expanding successful pilot projects into broader organizational applications.
• Customer Insights - Information about customer preferences and behaviors that can inform business strategies.
• Scientific Communication - The practice of conveying scientific information to various audiences effectively.
• Personalized Engagement - Tailoring interactions and communications to meet the specific needs and preferences of individual customers.
• AI-Driven Knowledge Extraction - The process of using AI to gather insights from large datasets.
• In Silico Screening - The use of computer simulations to predict the effectiveness of drug compounds.
• Regulatory Intelligence - The use of data and analytics to understand and comply with regulatory requirements.
• Patient Recruitment - The process of identifying and enrolling participants for clinical trials.
• Quality Control - The processes and procedures that ensure products meet specified requirements and standards.
ARTIFICIAL INTELLIGENCE PPT TEMPLATES
Source: Generative AI (GenAI) in the Pharmaceutical Industry PowerPoint (PPTX) Presentation Slide Deck, LearnPPT Consulting
Did you need more documents?
Consider a FlevyPro subscription from $39/month. View plans here.
For $10.00 more, you can download this document plus 2 more FlevyPro documents. That's just $13 each.