100+ Manufacturing Quality Assurance SOPs – Excel XLSX
Excel (XLSX)
BENEFITS OF THIS DOWNLOADABLE EXCEL DOCUMENT
- Provides a comprehensive system for ensuring consistent product quality and regulatory compliance across your manufacturing processes.
- Delivers a structured approach for managing inspections, testing, and process controls to reduce defects and improve production efficiency.
- Equips your team with standardized procedures to proactively address risks and ensure continuous improvement throughout the product lifecycle.
QUALITY MANAGEMENT EXCEL DESCRIPTION
Curated by McKinsey-trained Executives
🚨 100+ Manufacturing Quality Assurance SOPs Library 🚨
THE ULTIMATE TOOLKIT FOR STREAMLINING YOUR MANUFACTURING QUALITY ASSURANCE PROCESS AND ENSURING CONSISTENCY, COMPLIANCE, AND EXCELLENCE IN EVERY STEP OF PRODUCTION 💥
🔥 WHY MANUFACTURERS STRUGGLE TO MAINTAIN HIGH QUALITY STANDARDS AND ENSURE COMPLIANCE ACROSS THE BOARD
Let's face it:
Most manufacturers face major hurdles:
❌ Lack of standardized quality assurance procedures across teams and departments
❌ Insufficient testing protocols causing defects to go unnoticed
❌ Inefficient document control leading to confusion and errors
❌ Non-compliant processes risking major legal and regulatory issues
❌ Gaps in risk management preventing proactive issue resolution
❌ Delays due to poorly implemented inspection and process control systems
THE RESULT?
👉 Increased defects and product recalls
👉 Regulatory fines and compliance issues
👉 Wasted resources, increased production costs, and inefficiency
👉 Poor stakeholder confidence and loss of business
👉 Reduced product quality and compromised brand reputation
🚨 INTRODUCING: THE 150+ MANUFACTURING QUALITY ASSURANCE SOPs LIBRARY
This isn't just another set of generic procedures.
This is a:
✔ COMPREHENSIVE QUALITY MANAGEMENT SYSTEM
✔ END-TO-END PRODUCT INSPECTION AND TESTING FRAMEWORK
✔ PROCESS CONTROL AND OPTIMIZATION TOOL
✔ MATERIAL MANAGEMENT AND SUPPLY CHAIN ASSURANCE
✔ COMPLIANCE AND REGULATORY MANAGEMENT SOLUTION
💥 Stop leaving your product quality to chance.
👉 BUILD A SEAMLESS, COMPLIANT, AND HIGH-PERFORMANCE QUALITY MANAGEMENT SYSTEM.
📦 WHAT YOU GET
✔ 150+ Expertly Crafted Manufacturing Quality Assurance SOPs
✔ Fully Structured, Plug-and-Play Framework for Every Stage of Quality Management
✔ 15 Key Quality Assurance Clusters for Comprehensive Control
✔ Built for Quality Managers, Production Supervisors, Compliance Officers, and Inspectors
✔ Standardized Operating Procedures for Product Inspection, Testing, Process Control, and Compliance
✔ Designed to Maximize Product Quality, Reduce Defects, and Ensure Regulatory Compliance
🧠 EACH SOP FOLLOWS A UNIFIED ENTERPRISE STRUCTURE
Every SOP follows the SAME battle-tested framework:
🎯 Purpose
📌 Scope
👤 Owner / Role
📥 Inputs (Required Information & Documents)
⚙️ Process Steps (Detailed Workflow)
📤 Outputs / Deliverables
📊 KPIs / Success Metrics
⚠️ Risks / Controls
🔁 Review Frequency
📚 THE 150+ MANUFACTURING QUALITY ASSURANCE SOP LIBRARY
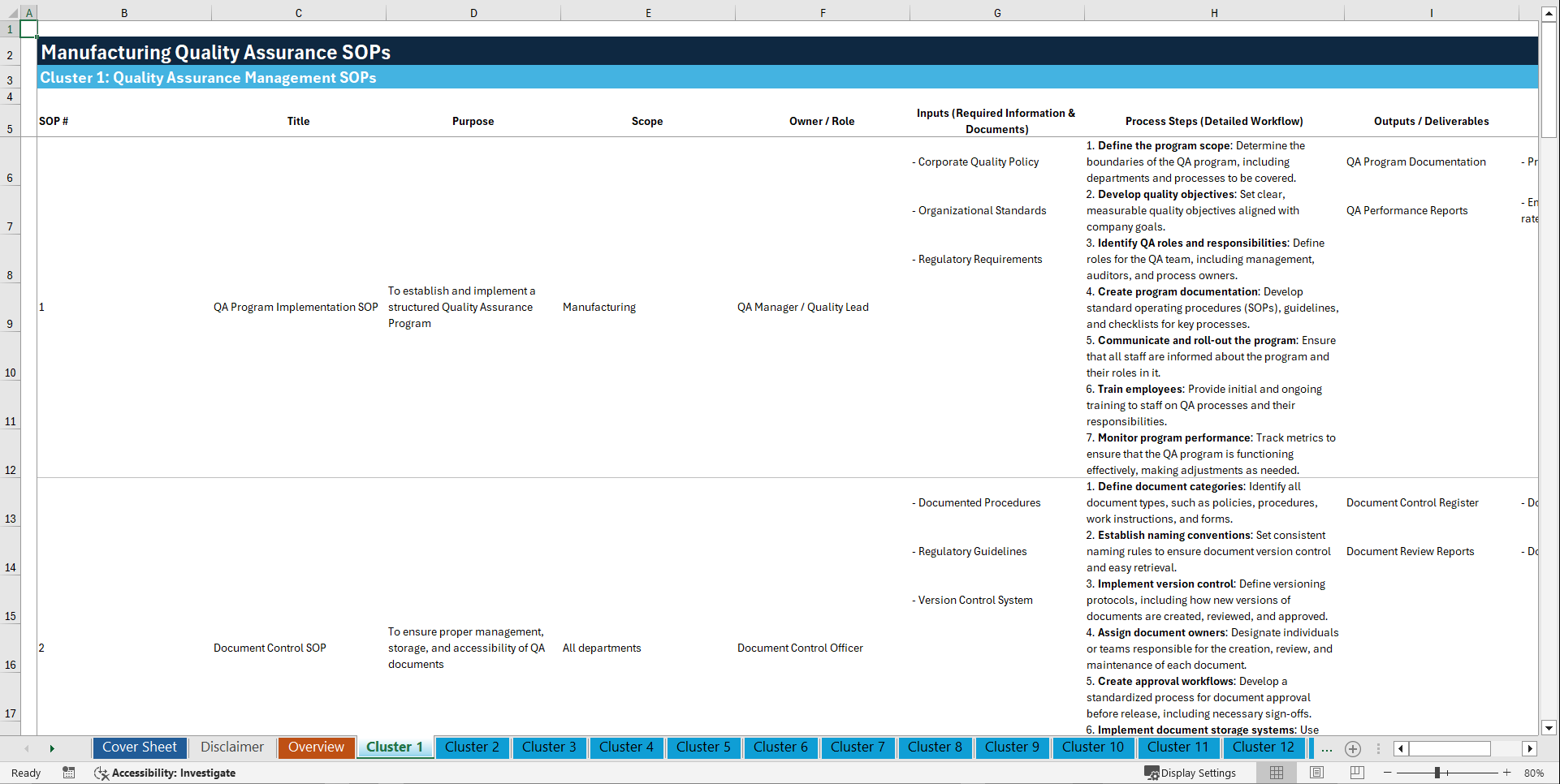
📊 CLUSTER 1: QUALITY ASSURANCE MANAGEMENT SOPs (1–10)
1. QA Program Implementation SOP
2. Document Control SOP
3. Internal Audit SOP
4. Corrective and Preventive Action (CAPA) SOP
5. Supplier Quality Management SOP
6. Quality Management System (QMS) SOP
7. Risk Management SOP
8. Training and Competency SOP
9. Audit and Inspection Procedure SOP
10. Non-Conformance Management SOP
📊 CLUSTER 2: PRODUCT INSPECTION AND TESTING SOPs (11–20)
11. Incoming Material Inspection SOP
12. In-Process Inspection SOP
13. Final Product Inspection SOP
14. Functional Testing SOP
15. Dimensional Testing SOP
16. Destructive Testing SOP
17. Non-Destructive Testing SOP
18. Analytical Testing SOP
19. Stability Testing SOP
20. Sampling Plan SOP
📊 CLUSTER 3: PROCESS CONTROL SOPs (21–30)
21. Process Validation SOP
22. Statistical Process Control SOP
23. Process Flow Control SOP
24. Equipment Calibration SOP
25. Machine Maintenance SOP
26. Failure Mode and Effects Analysis (FMEA) SOP
27. Control of Critical Process Parameters SOP
28. Continuous Improvement SOP
29. Production Line Setup and Monitoring SOP
30. Production Line Troubleshooting SOP
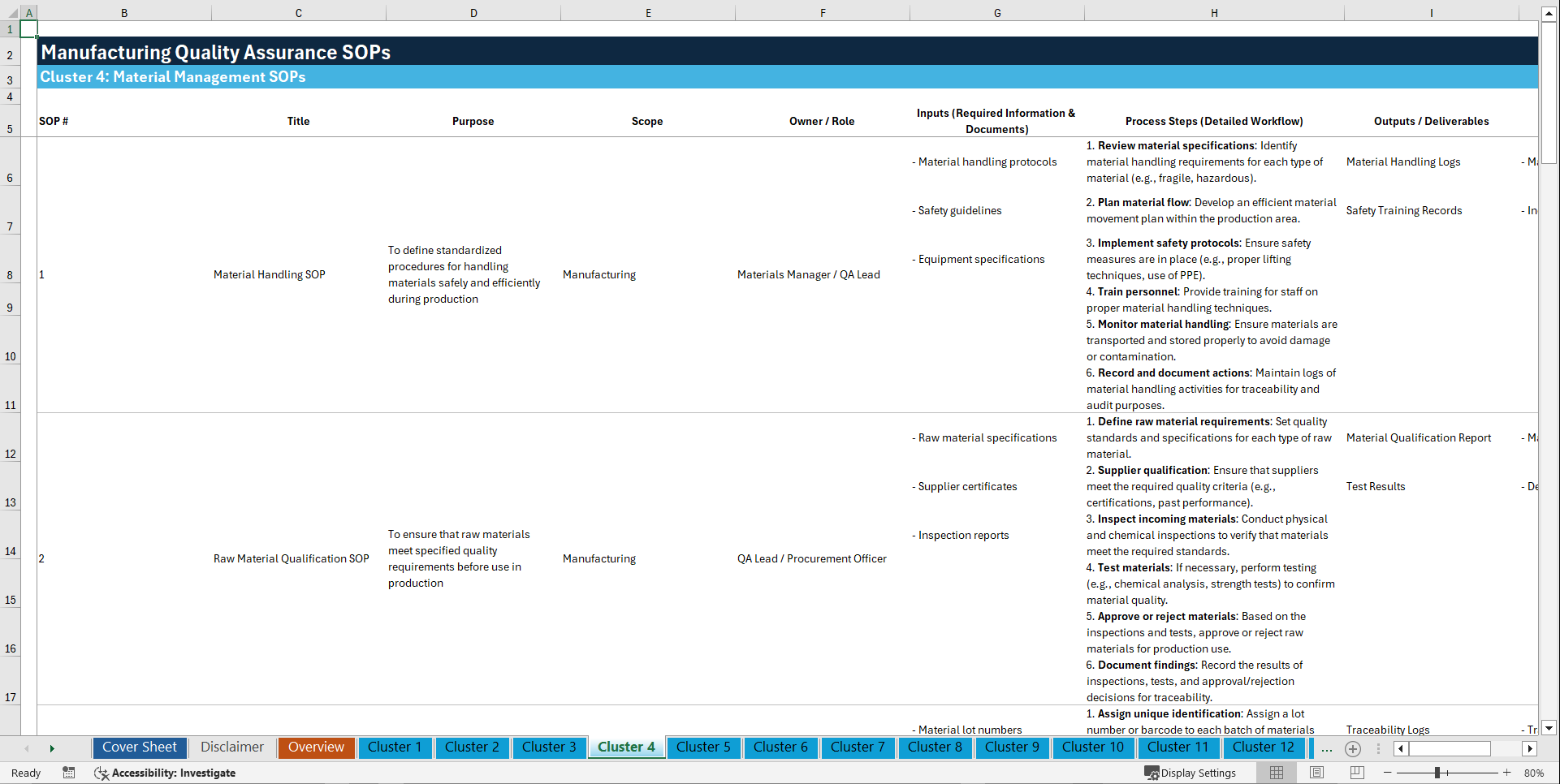
📊 CLUSTER 4: MATERIAL MANAGEMENT SOPs (31–40)
31. Material Handling SOP
32. Raw Material Qualification SOP
33. Material Traceability SOP
34. Materials Storage SOP
35. Rejection and Disposal of Defective Materials SOP
36. Hazardous Materials Handling SOP
37. Supplier Evaluation and Selection SOP
38. Lot Control SOP
39. Packaging Materials Quality Assurance SOP
40. Packaging and Storage Conditions SOP
📊 CLUSTER 5: COMPLIANCE AND REGULATORY SOPs (41–50)
41. FDA Compliance SOP
42. ISO 9001 Compliance SOP
43. GMP (Good Manufacturing Practice) Compliance SOP
44. Environmental Compliance SOP
45. Regulatory Inspection SOP
46. Product Recall SOP
47. Regulatory Documentation Management SOP
48. Auditing for Regulatory Compliance SOP
49. Environmental Health and Safety SOP
50. Product Labeling Compliance SOP
📊 CLUSTER 6: CALIBRATION AND INSTRUMENTATION SOPs (51–60)
51. Calibration Schedule Management SOP
52. Instrument Calibration SOP
53. Measurement System Analysis SOP
54. Calibration Documentation SOP
55. Equipment Qualification SOP
56. Calibration Equipment Maintenance SOP
57. Instrument Validation SOP
58. Control and Calibration of Test Equipment SOP
59. Monitoring and Validation of Test Instruments SOP
60. Calibration Failure Investigation SOP
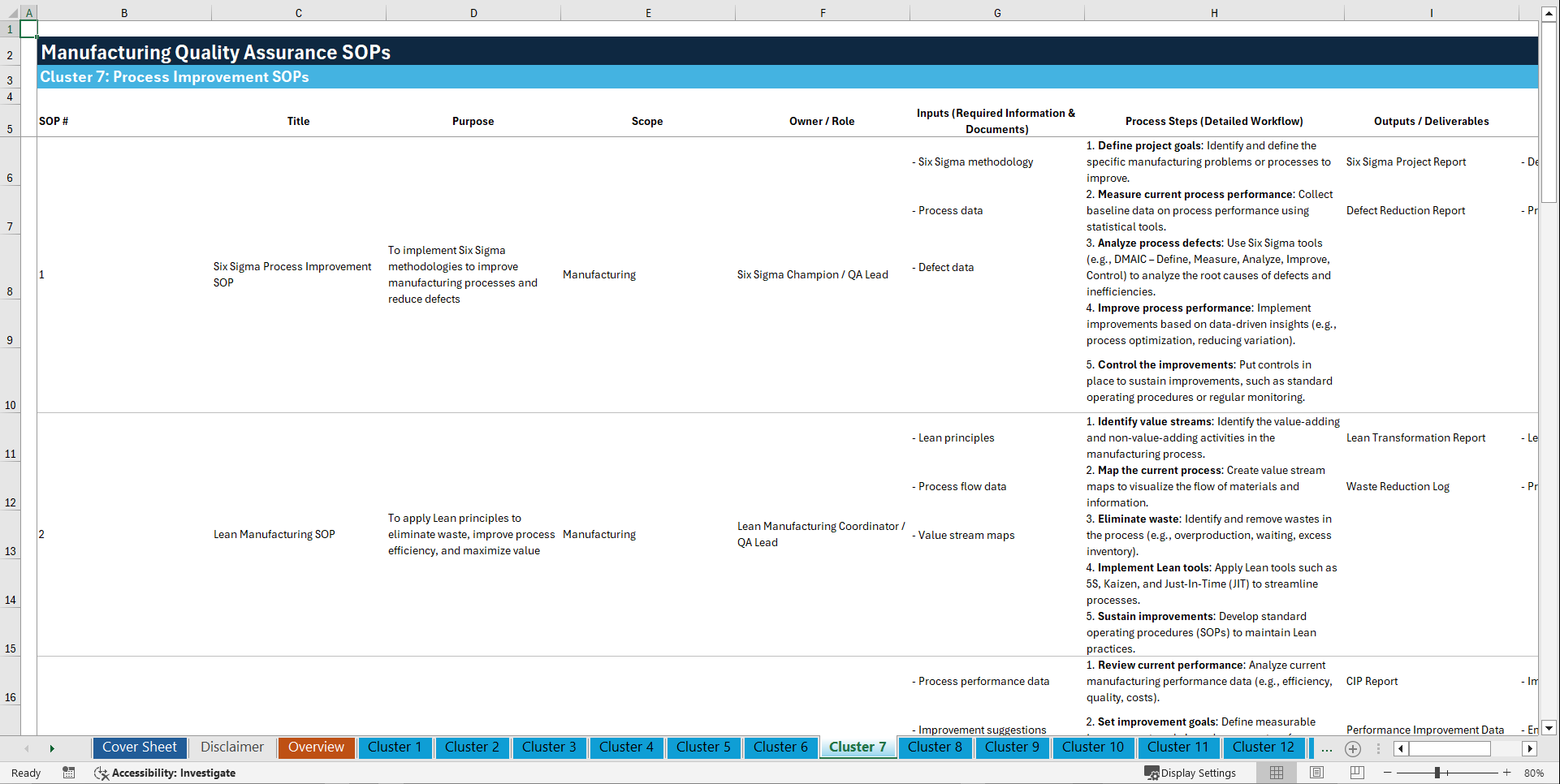
📊 CLUSTER 7: PROCESS IMPROVEMENT SOPs (61–70)
61. Six Sigma Process Improvement SOP
62. Lean Manufacturing SOP
63. Continuous Improvement Process (CIP) SOP
64. Kaizen Event SOP
65. Root Cause Analysis SOP
66. Corrective Action Implementation SOP
67. Performance Metrics and KPIs SOP
68. Problem-Solving Methodology SOP
69. Waste Reduction SOP
70. Value Stream Mapping SOP
📊 CLUSTER 8: PRODUCT LIFECYCLE MANAGEMENT SOPs (71–80)
71. New Product Development SOP
72. Design Control SOP
73. Design Verification and Validation SOP
74. Change Control SOP
75. Prototype Testing SOP
76. Product Risk Assessment SOP
77. Product Lifecycle Documentation SOP
78. Configuration Management SOP
79. Design Review SOP
80. End-of-Life Product Disposition SOP
📊 CLUSTER 9: PACKAGING AND LABELING SOPs (81–90)
81. Packaging Design and Development SOP
82. Labeling Control SOP
83. Packaging Material Inspection SOP
84. Packaging Process Validation SOP
85. Sterility Assurance SOP
86. Final Packaging Inspection SOP
87. Packaging and Storage Conditions SOP
88. Labeling Compliance SOP
89. Traceability and Serialization SOP
90. Packaging Process Monitoring SOP
📊 CLUSTER 10: ENVIRONMENTAL AND SAFETY MANAGEMENT SOPs (91–100)
91. Hazardous Waste Management SOP
92. Personal Protective Equipment (PPE) SOP
93. Environmental Monitoring SOP
94. Safety Audit SOP
95. Incident Reporting and Investigation SOP
96. Fire Safety and Emergency Response SOP
97. Ergonomics and Safety SOP
98. Energy Management SOP
99. Occupational Health and Safety SOP
100. Chemical Handling SOP
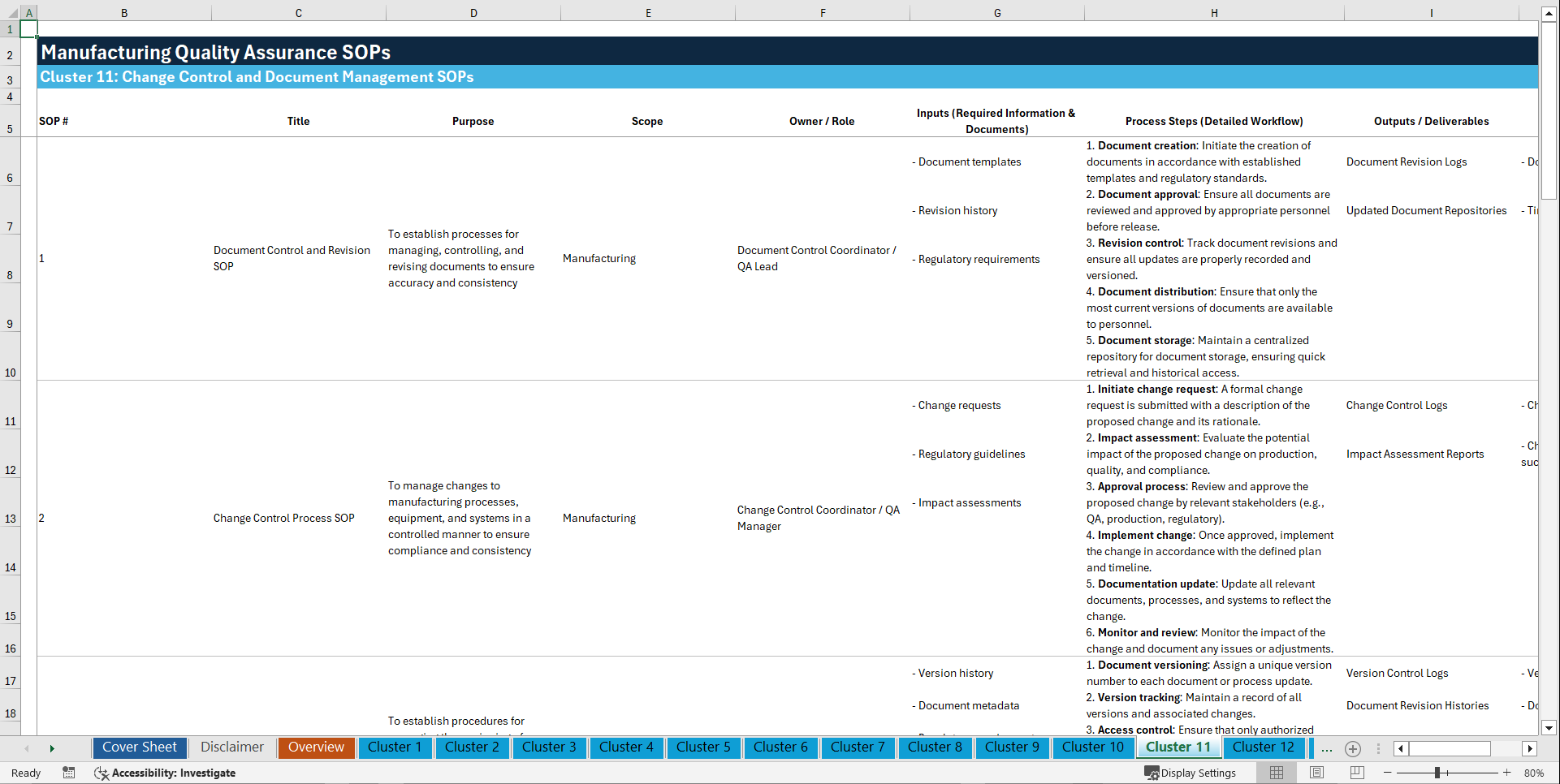
📊 CLUSTER 11: CHANGE CONTROL AND DOCUMENT MANAGEMENT SOPs (101–110)
101. Document Control and Revision SOP
102. Change Control Process SOP
103. Version Control SOP
104. Document Retention SOP
105. Configuration Management SOP
106. Electronic Document Management SOP
107. Document Distribution SOP
108. Obsolete Document Management SOP
109. Software Validation SOP
110. Quality Control Records Management SOP
📊 CLUSTER 12: SUPPLIER MANAGEMENT AND AUDITING SOPs (111–120)
111. Supplier Auditing SOP
112. Supplier Qualification SOP
113. Supplier Performance Evaluation SOP
114. Supplier Non-Conformance Management SOP
115. Supplier Relationship Management SOP
116. Vendor Qualification SOP
117. Vendor Quality Assurance SOP
118. Subcontractor Management SOP
119. Outsourced Process Control SOP
120. Supplier Corrective Action Response SOP
📊 CLUSTER 13: CUSTOMER COMPLAINT AND FEEDBACK SOPs (121–130)
121. Customer Complaint Handling SOP
122. Feedback Collection and Analysis SOP
123. Corrective Action for Customer Complaints SOP
124. Customer Satisfaction Measurement SOP
125. Customer Communication Protocol SOP
126. Root Cause Analysis for Complaints SOP
127. Escalation Procedures SOP
128. Product Return Management SOP
129. Warranty Claims SOP
130. Product Feedback Loop SOP
📊 CLUSTER 14: PRODUCTION AND ASSEMBLY SOPs (131–140)
131. Production Scheduling SOP
132. Batch Manufacturing SOP
133. Cleanroom Protocol SOP
134. Assembly Line Quality Control SOP
135. Operator Training SOP
136. Production Equipment Setup SOP
137. Equipment Qualification SOP
138. Production Line Troubleshooting SOP
139. End-of-Line Quality Control SOP
140. Process Monitoring SOP
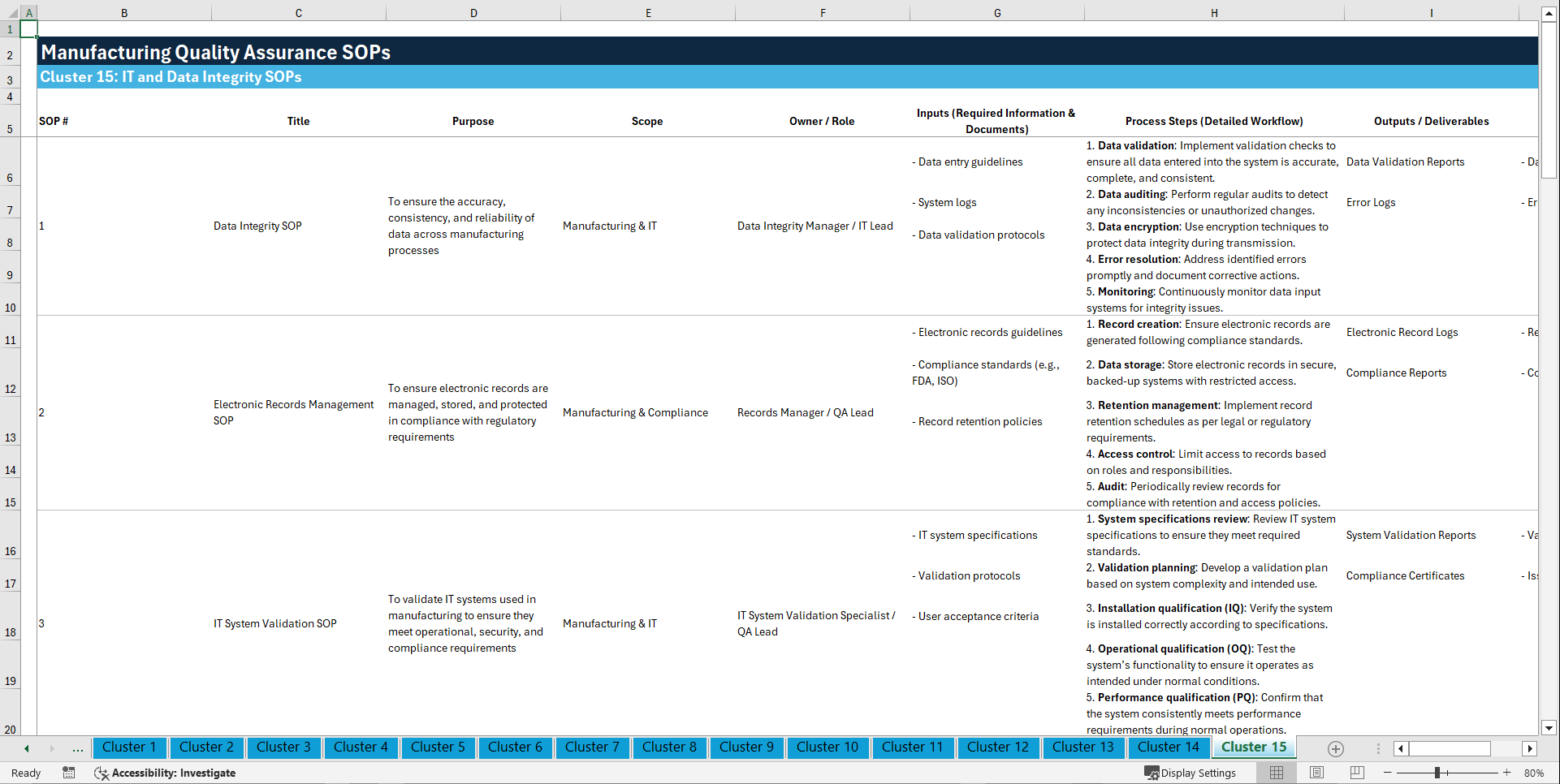
📊 CLUSTER 15: IT AND DATA INTEGRITY SOPs (141–150)
141. Data Integrity SOP
142. Electronic Records Management SOP
143. IT System Validation SOP
144. Cybersecurity in Manufacturing SOP
145. Backup and Recovery SOP
146. Software System Validation SOP
147. User Access Control SOP
148. Data Integrity and Audit Trail SOP
149. Software Lifecycle Management SOP
150. Cloud Computing
in Manufacturing SOP
💥 WHAT THIS SYSTEM ACTUALLY DOES
✔ Streamlines your quality assurance and process control across manufacturing operations
✔ Provides a structured approach for managing inspections, testing, and material control
✔ Ensures the highest levels of product quality and compliance with industry standards
✔ Aligns your team with transparent procedures and consistent quality metrics
✔ Mitigates risks through proactive auditing, CAPA, and corrective actions
✔ Enhances process efficiency through continuous improvement and lean manufacturing strategies
🧠 WHO THIS IS BUILT FOR
✔ Quality Assurance Managers
✔ Compliance Officers
✔ Production Supervisors and Line Workers
✔ Inspectors and Test Engineers
✔ Process Control Specialists
✔ Risk Management and Safety Officers
✔ Regulatory and Environmental Compliance Teams
⚡ FINAL WORD
Manufacturers often fail to maintain quality standards because:
👉 LACK OF STANDARDIZED AND COMPREHENSIVE QA PROCEDURES
👉 INSUFFICIENT INSPECTION AND TESTING SYSTEMS
👉 COMPLIANCE AND REGULATORY RISKS DUE TO INCONSISTENCY
👉 INEFFECTIVE RISK MANAGEMENT AND PROCESS CONTROL
The 150+ Manufacturing Quality Assurance SOPs Library solves all of these problems and more.
🚨 STOP LEAVING YOUR QUALITY CONTROL TO CHANCE. START BUILDING A HIGH-PERFORMANCE, COMPLIANT, AND EFFICIENT QUALITY ASSURANCE SYSTEM TODAY. 🚨
Key Words:
Strategy & Transformation, Growth Strategy, Strategic Planning, Strategy Frameworks, Innovation Management, Pricing Strategy, Core Competencies, Strategy Development, Business Transformation, Marketing Plan Development, Product Strategy, Breakout Strategy, Competitive Advantage, Mission, Vision, Values, Strategy Deployment & Execution, Innovation, Vision Statement, Core Competencies Analysis, Corporate Strategy, Product Launch Strategy, BMI, Blue Ocean Strategy, Breakthrough Strategy, Business Model Innovation, Business Strategy Example, Corporate Transformation, Critical Success Factors, Customer Segmentation, Customer Value Proposition, Distinctive Capabilities, Enterprise Performance Management, KPI, Key Performance Indicators, Market Analysis, Market Entry Example, Market Entry Plan, Market Intelligence, Market Research, Market Segmentation, Market Sizing, Marketing, Michael Porter's Value Chain, Organizational Transformation, Performance Management, Performance Measurement, Platform Strategy, Product Go-to-Market Strategy, Reorganization, Restructuring, SWOT, SWOT Analysis, Service 4.0, Service Strategy, Service Transformation, Strategic Analysis, Strategic Plan Example, Strategy Deployment, Strategy Execution, Strategy Frameworks Compilation, Strategy Methodologies, Strategy Report Example, Value Chain, Value Chain Analysis, Value Innovation, Value Proposition, Vision Statement, Corporate Strategy, Business Development, Business plan pdf, business plan, PDF, Business Plan DOC, Business Plan Template, PPT, Market strategy playbook, strategic market planning, competitive analysis tools, market segmentation frameworks, growth strategy templates, product positioning strategy, market execution toolkit, strategic alignment playbook, KPI and OKR frameworks, business growth strategy guide, cross-functional strategy templates, market risk management, market strategy PowerPoint doc, guide, ebook, e-book ,McKinsey Change Playbook, Organizational change management toolkit, Change management frameworks 2025, Influence model for change, Change leadership strategies, Behavioral change in organizations, Change management PowerPoint templates, Transformational leadership in change, supply chain KPIs, supply chain KPI toolkit, supply chain PowerPoint template, logistics KPIs, procurement KPIs, inventory management KPIs, supply chain performance metrics, manufacturing KPIs, supply chain dashboard, supply chain strategy KPIs, reverse logistics KPIs, sustainability KPIs in supply chain, financial supply chain KPIs, warehouse KPIs, digital supply chain KPIs, 1200 KPIs, supply chain scorecard, KPI examples, supply chain templates, Corporate Finance SOPs, Finance SOP Excel Template, CFO Toolkit, Finance Department Procedures, Financial Planning SOPs, Treasury SOPs, Accounts Payable SOPs, Accounts Receivable SOPs, General Ledger SOPs, Accounting Policies Template, Internal Controls SOPs, Finance Process Standardization, Finance Operating Procedures, Finance Department Excel Template, FP&A Process Documentation, Corporate Finance Template, Finance SOP Toolkit, CFO Process Templates, Accounting SOP Package, Tax Compliance SOPs, Financial Risk Management Procedures.
NOTE: Our digital products are sold on an "as is" basis, making returns and refunds unavailable post-download. Please preview and inquire before purchasing. Please contact us before purchasing if you have any questions! This policy aligns with the standard Flevy Terms of Usage.
Got a question about the product? Email us at support@flevy.com or ask the author directly by using the "Ask the Author a Question" form. If you cannot view the preview above this document description, go here to view the large preview instead.
Source: Best Practices in Quality Management Excel: 100+ Manufacturing Quality Assurance SOPs Excel (XLSX) Spreadsheet, SB Consulting