Biotechnology Value Chain – PowerPoint PPTX Template
PowerPoint (PPTX) 31 Slides
BENEFITS OF THIS DOWNLOADABLE POWERPOINT DOCUMENT
- Provides a framework for examining the Biotech Industry.
- Identifies 15 important insights and considerations for Biotech Value Chain Analysis.
- Provides a detailed breakdown of the impact of Digital Transformation across the Biotech Value Chain.
VALUE CHAIN ANALYSIS PPT TEMPLATE DESCRIPTION
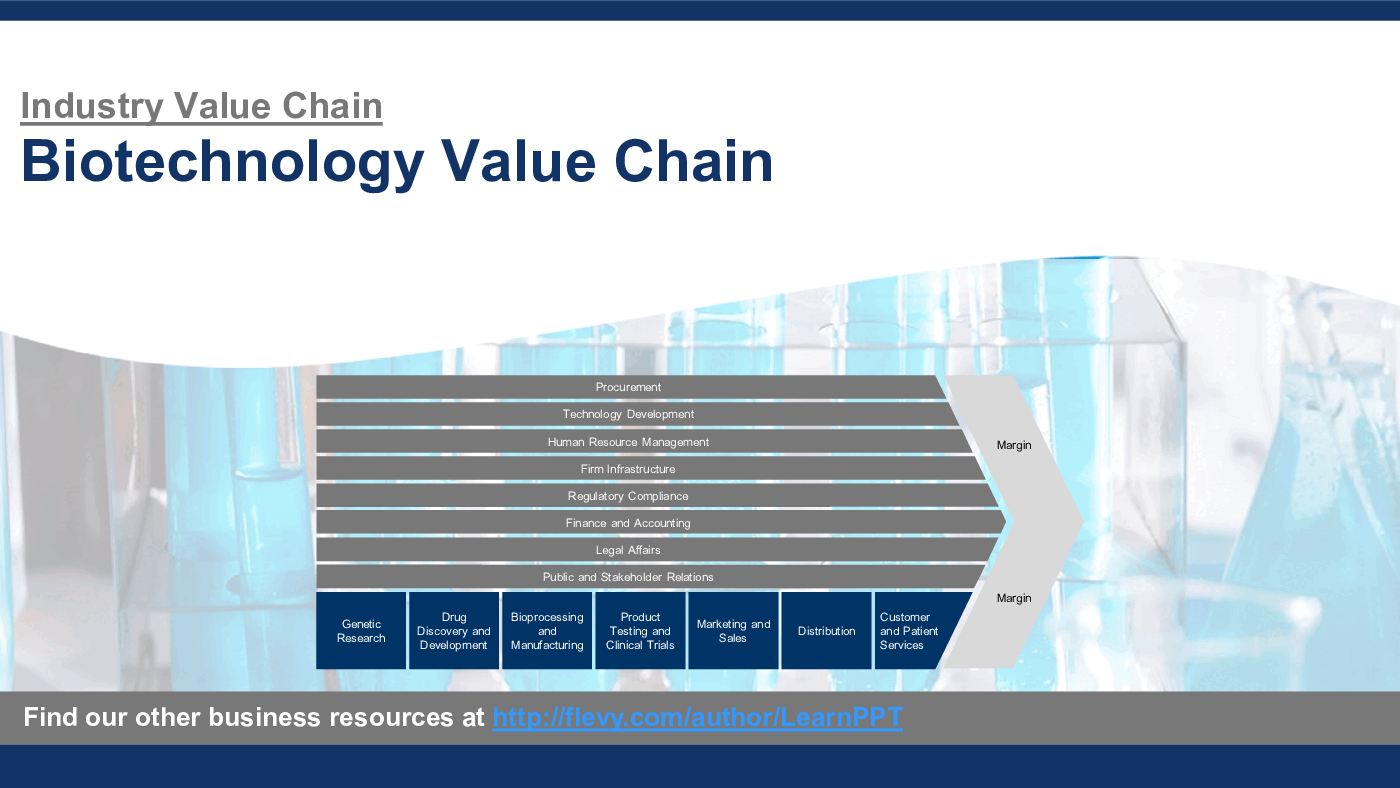
An Industry Value Chain is a visual representation of the series of steps an organization in a specific industry takes to deliver a product or service to the market. It captures the main business functions and processes that are involved in delivering the end product or service, illustrating how each step adds value and contributes to competitive advantage. It often encompasses both primary and support activities.
This PowerPoint presentation captures the Biotech Industry Value Chain, which includes the following primary and support activities:
Primary Activities
1. Genetic Research
2. Drug Discovery and Development
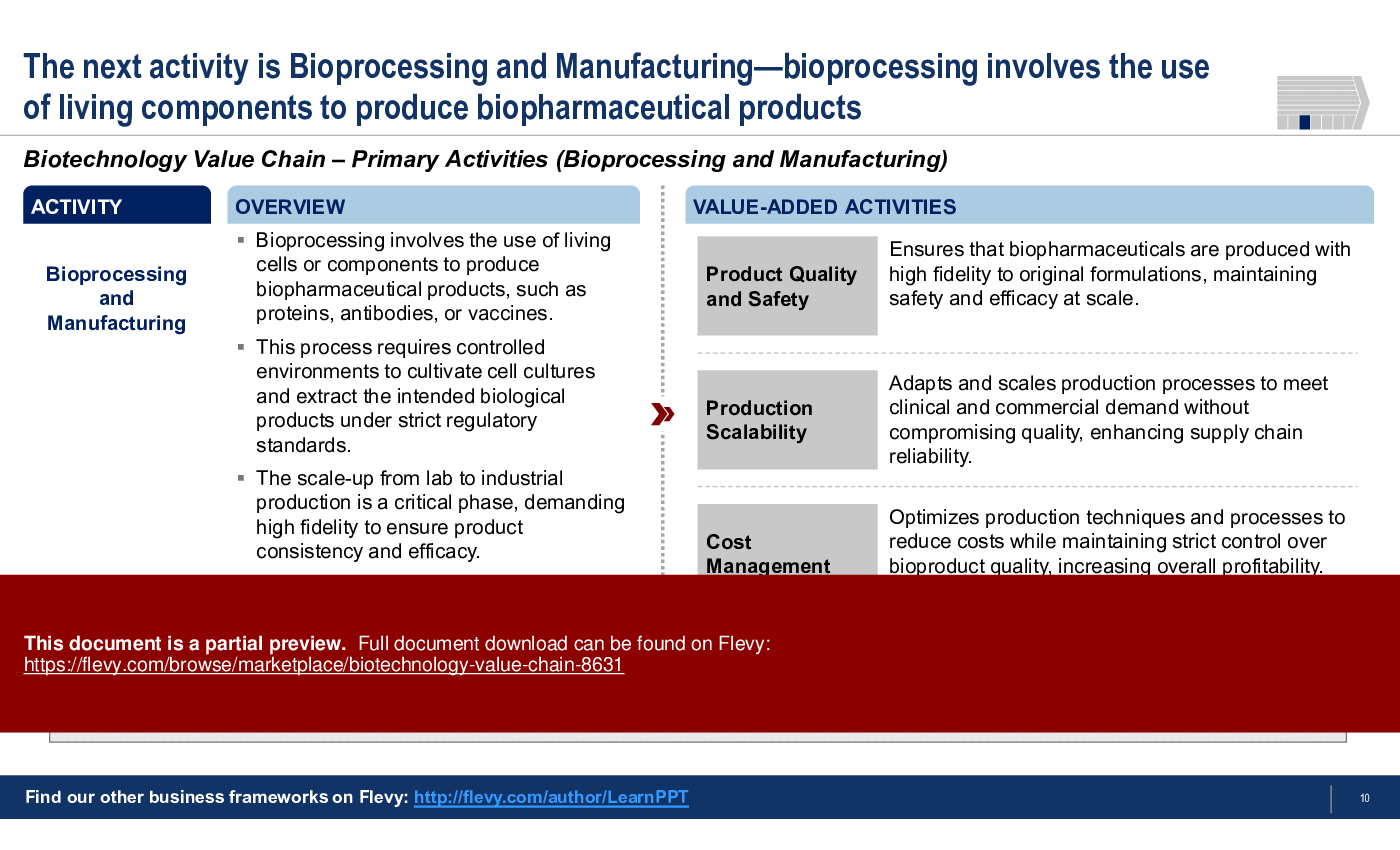
3. Bioprocessing and Manufacturing
4. Product Testing and Clinical Trials
5. Marketing and Sales
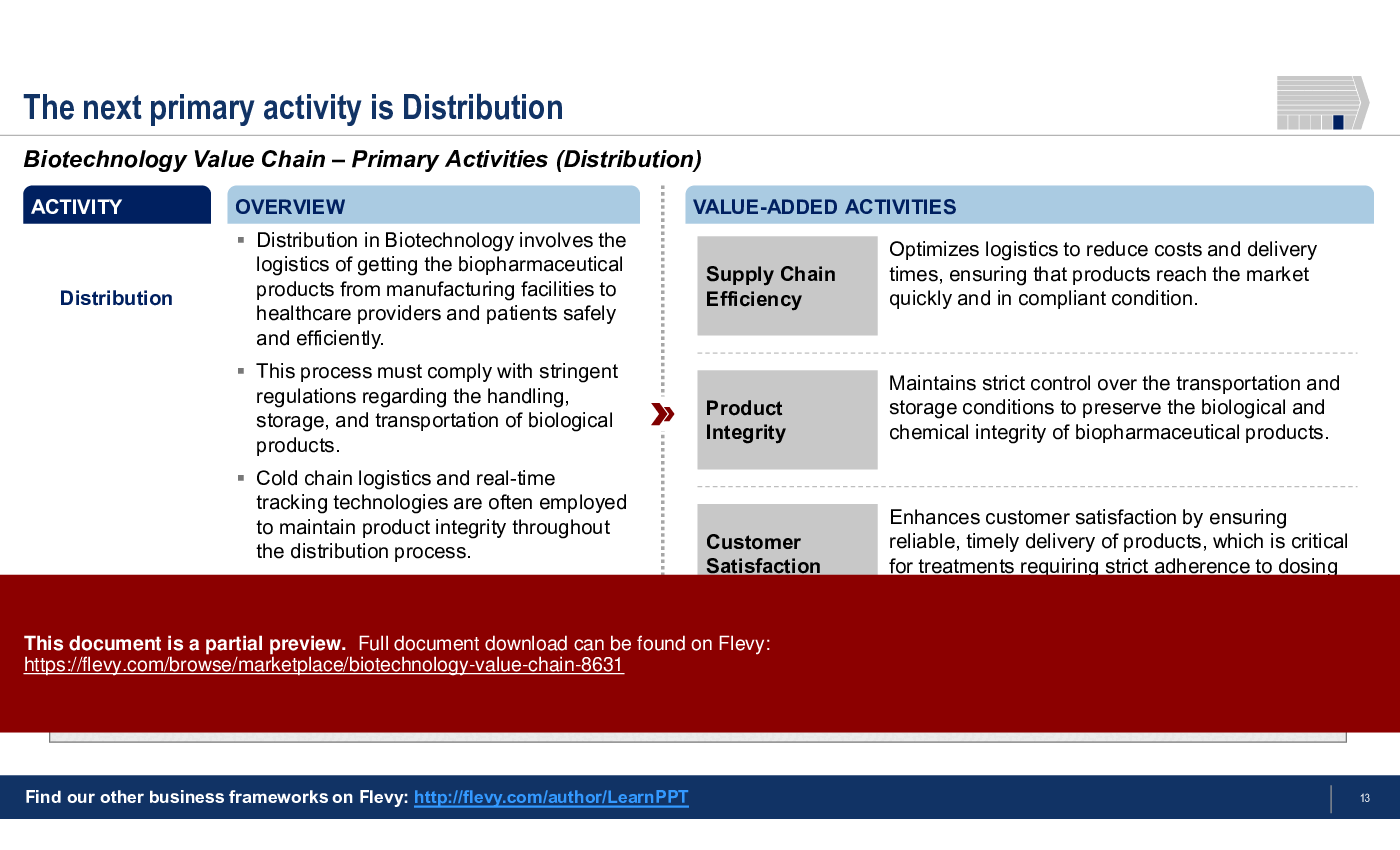
6. Distribution
7. Customer and Patient Services
Support Activities
1. Procurement
2. Technology Development
3. Human Resource Management
4. Firm Infrastructure
5. Regulatory Compliance
6. Finance and Accounting
7. Legal Affairs
8. Public and Stakeholder Relations
These activities form the core framework for analyzing and organizing the operations within the Biotech Industry, particularly for companies engaged in cutting-edge fields like genetic research and personalized medicine. The Biotechnology Value Chain is intricately designed to foster innovation from genetic research through to the delivery of personalized medicine, integrating advanced scientific knowledge with stringent regulatory oversight.
This PowerPoint presentation dives deeper into each of these activities, highlighting key elements. This presentation also discusses Biotechnology Value Chain Analysis, highlighting a multitude of key considerations and potential insights to pay attention to. We further discuss the significant impact of Digital Transformation and various specific emergent technologies on the Biotech Industry.
The Biotechnology Value Chain is intricately designed to foster innovation from genetic research through to the delivery of personalized medicine, integrating advanced scientific knowledge with stringent regulatory oversight. Efficient integration of each stage in the Biotech Value Chain is critical for accelerating the development of new therapies and ensuring they are both efficacious and safe for patient use. Strategic partnerships across academia, industry, and regulatory bodies within the Biotech Value Chain can significantly shorten development timelines and enhance market entry success.
Got a question about the product? Email us at support@flevy.com or ask the author directly by using the "Ask the Author a Question" form. If you cannot view the preview above this document description, go here to view the large preview instead.
PRESENTATION DEEP DIVE ANALYSIS
This deep-dive analysis was generated from the full 31-slide PowerPoint presentation.
Executive Summary
The Biotechnology Value Chain presentation offers a detailed framework for understanding the essential steps that organizations in the biotech industry undertake to deliver innovative products and services. Crafted by former consultants from leading firms like McKinsey and BCG, this consulting-grade resource provides a comprehensive analysis of both primary and support activities within the biotech sector. Buyers will gain insights into optimizing operations from genetic research to personalized medicine delivery, ensuring compliance with regulatory standards while fostering innovation. This framework is designed to enhance strategic decision-making and operational efficiency in a highly competitive market.
Who This Is For and When to Use
• Biotechnology executives overseeing product development and market strategy
• R&D leaders focused on genetic research and drug discovery
• Operations managers involved in bioprocessing and manufacturing
• Marketing teams responsible for product promotion and sales strategies
• Compliance officers ensuring adherence to regulatory standards
• Financial analysts managing investments in biotech projects
Best-fit moments to use this deck:
• During strategic planning sessions to align operational goals with market needs
• When assessing the efficiency of the biotech value chain for potential improvements
• In workshops aimed at fostering innovation and collaboration across departments
• For training sessions on regulatory compliance and market entry strategies
Learning Objectives
• Define the components of the Biotechnology Value Chain and their interdependencies
• Analyze the role of genetic research in developing targeted therapies
• Evaluate the efficiency of drug discovery and development processes
• Assess the impact of bioprocessing and manufacturing on product quality
• Identify best practices in marketing and sales within the biotech sector
• Develop strategies for effective distribution and customer engagement
• Understand the regulatory landscape affecting biotech operations
• Explore the importance of stakeholder relations in enhancing market acceptance
Table of Contents
• Executive Summary (page 3)
• Biotechnology Value Chain (page 4)
• Primary Activities (page 5)
• Support Activities (page 6)
• Biotechnology Value Chain Analysis (page 24)
Primary Topics Covered
• Genetic Research - Focuses on studying genes to identify disease causes and develop genetic profiles, utilizing techniques like CRISPR and bioinformatics.
• Drug Discovery and Development - Involves identifying biological compounds to create therapeutic products, emphasizing collaboration with research institutions.
• Bioprocessing and Manufacturing - Utilizes living cells to produce biopharmaceuticals, ensuring compliance with regulatory standards during production.
• Product Testing and Clinical Trials - Evaluates the safety and efficacy of products through systematic studies, critical for regulatory approvals.
• Marketing and Sales - Promotes biopharmaceutical products to healthcare providers and patients, requiring clear communication of benefits and risks.
• Distribution - Manages logistics to ensure safe and efficient delivery of products, adhering to stringent regulations.
• Customer and Patient Services - Supports patients post-sale, enhancing adherence and satisfaction through education and feedback mechanisms.
Deliverables, Templates, and Tools
• Framework for analyzing the Biotechnology Value Chain
• Templates for conducting value chain assessments
• Guidelines for regulatory compliance documentation
• Checklists for product testing and clinical trial management
• Marketing strategy templates tailored for biotech products
• Customer engagement plans focusing on patient education
Slide Highlights
• Overview of the Biotechnology Value Chain with primary and support activities
• Detailed breakdown of genetic research techniques and their applications
• Insights into drug discovery processes and the role of AI in enhancing efficiency
• Visual representation of bioprocessing and manufacturing workflows
• Key considerations for regulatory compliance and market access strategies
Potential Workshop Agenda
Biotechnology Value Chain Overview (60 minutes)
• Introduce the concept of the value chain and its significance
• Discuss primary and support activities in detail
• Explore case studies of successful biotech companies
Regulatory Compliance Strategies (45 minutes)
• Review the regulatory landscape affecting biotech operations
• Identify best practices for compliance and risk management
• Engage in a Q&A session to address specific concerns
Customer Engagement and Marketing (60 minutes)
• Analyze effective marketing strategies for biotech products
• Discuss the importance of customer feedback in product development
• Develop action plans for enhancing patient services
Customization Guidance
• Tailor the framework to reflect specific organizational goals and market dynamics
• Update regulatory compliance sections to align with current laws and standards
• Incorporate company-specific case studies to enhance relevance
• Adjust marketing strategies based on target audience insights and feedback
Secondary Topics Covered
• The impact of digital transformation on the biotech industry
• Strategies for effective stakeholder engagement
• Trends in personalized medicine and genetic therapies
• Challenges in supply chain management for biotech products
• Innovations in bioprocessing technologies
Topic FAQ
What are the main stages of a biotechnology value chain I should map?
A biotech value chain typically maps 7 primary stages: genetic research; drug discovery and development; bioprocessing and manufacturing; product testing and clinical trials; marketing and sales; distribution; and customer and patient services. These stages are the core primary activities described in the value-chain framework.How does genetic research contribute to personalized medicine in the value chain?
Genetic research identifies disease causes and genetic profiles that enable targeted therapies and personalized medicine. Techniques cited include CRISPR and bioinformatics, which feed findings into downstream drug discovery, development and clinical testing processes to support personalized treatment approaches using specific genetic data.What role does regulatory compliance play across the biotechnology value chain?
Regulatory compliance is a named support activity that ensures production, testing, and market access meet legal standards. It must be integrated across stages—bioprocessing, clinical trials, distribution and marketing—to secure approvals and maintain operational legitimacy, and the framework includes guidelines for compliance documentation.How can digital transformation change operations within the biotech value chain?
Digital transformation can streamline processes, enhance data analysis, and improve patient engagement across research, development, manufacturing and post-market services. The framework covers digital transformation as a secondary topic, highlighting how technologies affect innovation, data workflows and stakeholder interactions within the value chain.What should I look for when choosing a biotech value chain toolkit for my team?
Look for slide-based frameworks and practical templates that map primary and support activities, regulatory guidance, clinical trial checklists, marketing templates and customer engagement plans. Also check for customization guidance and workshop-ready agendas; practical inclusions such as checklists for clinical trials and regulatory compliance templates are useful.How much time and what session formats support applying a biotech value chain framework?
The framework is designed for use in typical workshop formats and training modules—examples include a 60-minute value chain overview, a 45-minute regulatory compliance session, and a 60-minute customer engagement module—enabling focused team alignment and targeted follow-up work in those specified time blocks.What is the cost/value trade-off of buying templated biotech frameworks versus building from scratch?
Purchasing templates provides pre-built assessment tools, regulatory documentation guidelines and checklists that reduce upfront design time and standardize analysis. The referenced product is a 31-slide PowerPoint that bundles templates for value chain assessments, regulatory guidelines, clinical trial checklists and marketing templates as ready-to-use assets.I need to restructure R&D after a merger—how should I map capabilities?
Use value-chain mapping to document capabilities across genetic research, drug discovery, and bioprocessing, then overlay support activities like technology development, regulatory compliance and talent management to identify gaps. The biotechnology value chain framework organizes primary activities such as genetic research, drug discovery and bioprocessing for this purpose.How can marketing teams use value chain analysis to improve a biotech product launch?
Marketing teams can use the value-chain perspective to align messaging, distribution and patient services with clinical evidence and regulatory parameters, plan provider and patient outreach, and develop customer education programs. Practical artifacts include marketing strategy templates and customer engagement plans to support launch activities.What tools support clinical trial management within a biotech value chain analysis?
Clinical trial management is supported by checklists for product testing and clinical trials and by regulatory compliance documentation guidelines. These tools help standardize trial procedures, oversight and approvals; the framework explicitly lists checklists for clinical trial management as a deliverable.Document FAQ
These are questions addressed within this presentation.
What is the Biotechnology Value Chain?
The Biotechnology Value Chain outlines the series of steps organizations take to deliver biotech products, from genetic research to market delivery, highlighting both primary and support activities.
How can this framework improve operational efficiency?
By analyzing each component of the value chain, organizations can identify inefficiencies and optimize processes, leading to faster product development and enhanced market responsiveness.
What role does regulatory compliance play in the value chain?
Regulatory compliance ensures that all activities meet stringent standards, which is crucial for gaining market access and maintaining operational legitimacy.
How can digital transformation impact the biotech value chain?
Digital technologies can streamline processes, enhance data analysis, and improve patient engagement, ultimately leading to more efficient operations and innovative product development.
What are the primary activities in the Biotechnology Value Chain?
The primary activities include genetic research, drug discovery and development, bioprocessing and manufacturing, product testing and clinical trials, marketing and sales, distribution, and customer and patient services.
What are the support activities in the Biotechnology Value Chain?
Support activities encompass procurement, technology development, human resource management, firm infrastructure, regulatory compliance, finance and accounting, legal affairs, and public and stakeholder relations.
How can organizations leverage this framework for strategic planning?
Organizations can use the framework to align their operational goals with market needs, ensuring that each component of the value chain contributes to overall business objectives.
What are the key considerations for conducting a Biotechnology Value Chain Analysis?
Key considerations include assessing technological innovation, regulatory compliance, intellectual property management, R&D efficiency, funding allocation, and stakeholder engagement.
Glossary
• Biotechnology Value Chain - A framework outlining the steps in delivering biotech products, from research to market.
• Genetic Research - The study of genes to understand diseases and develop therapies.
• Drug Discovery - The process of identifying compounds that can treat diseases.
• Bioprocessing - The use of living cells to produce biological products.
• Clinical Trials - Systematic studies to evaluate the safety and efficacy of products.
• Regulatory Compliance - Adherence to laws and regulations governing biotech operations.
• Stakeholder Engagement - Building relationships with parties interested in the biotech company's activities.
• Digital Transformation - The integration of digital technologies to enhance operations and innovation.
• Market Access - Strategies to enter and succeed in target markets.
• Customer Engagement - Interactions with patients and healthcare providers to enhance satisfaction and adherence.
• Supply Chain Management - The oversight of materials and products from production to delivery.
• Intellectual Property - Legal rights protecting innovations and inventions in biotech.
• Funding Allocation - The distribution of financial resources to support R&D and operations.
• Product Lifecycle Management - Managing a product's journey from development to market withdrawal.
• Personalized Medicine - Tailoring treatments to individual patient needs based on genetic information.
• Quality Control - Ensuring products meet safety and efficacy standards throughout production.
• Talent Management - Strategies for attracting and retaining skilled professionals in biotech.
• Public Relations - Managing the company's reputation and communication with external parties.
• Crisis Management - Strategies for handling potential public relations challenges.
• Operational Efficiency - Streamlining processes to enhance productivity and responsiveness.
VALUE CHAIN ANALYSIS PPT TEMPLATES
Source: Best Practices in Value Chain Analysis, Healthcare, Life Sciences, Biotech PowerPoint Slides: Biotechnology Value Chain PowerPoint (PPTX) Presentation Slide Deck, LearnPPT Consulting