ISO 13485:2016 Medical Devices - Awareness Training (PowerPoint PPTX Slide Deck)
PowerPoint (PPTX) 152 Slides
BENEFITS OF THIS POWERPOINT DOCUMENT
- Empowers professionals with comprehensive knowledge and tools to navigate the intricate landscape of ISO standards and medical device quality management.
- Equips organizations with actionable insights and practical strategies for implementing ISO 13485:2016 effectively, driving continuous improvement and regulatory compliance.
- Guides stakeholders through real-world case studies, enabling them to glean insights from successful implementations and overcome challenges in achieving ISO 13485:2016 certification.
HEALTHCARE PPT DESCRIPTION
Curated by McKinsey-trained Executives
Unlocking Success in Medical Device Quality Management with ISO 13485:2016 Awareness Training Toolkit
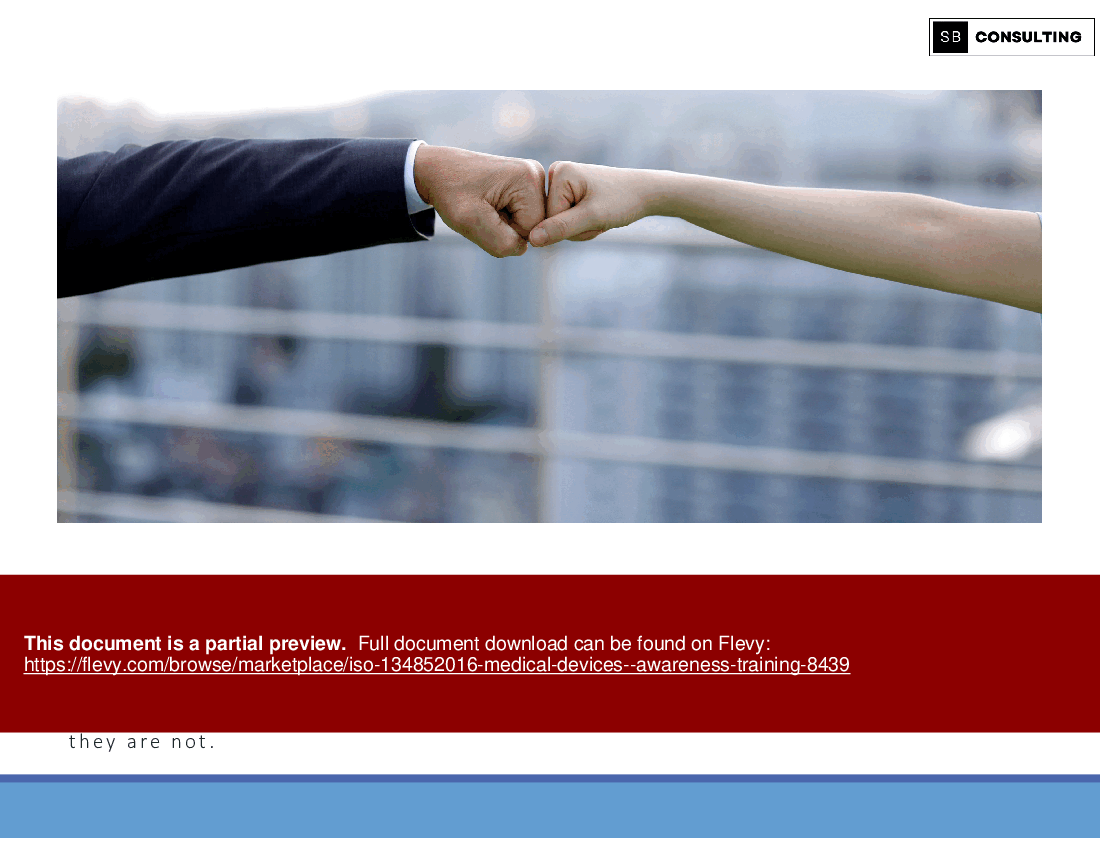
Welcome to a comprehensive journey into the world of ISO 13485:2016, the gold standard for quality management systems in the medical device industry. Whether you're a seasoned professional or new to the field, our ISO 13485 Awareness Training Toolkit is your essential companion for navigating the complexities of medical device quality management.
CONTENT OVERVIEW
• What is ISO
• About ISO
• What are Standards
• Medical Devices Definition
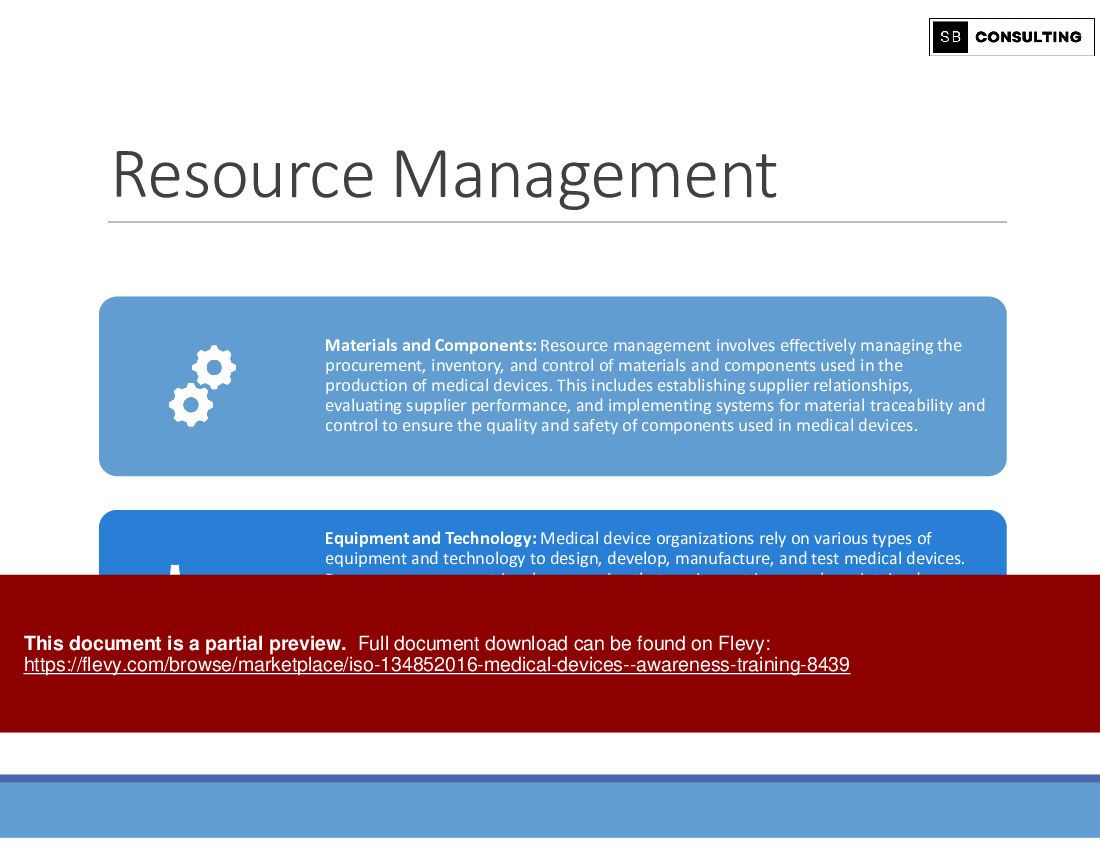
• Resource Management
• What is ISO 13485:2016
• Benefits of ISO 13485:2016
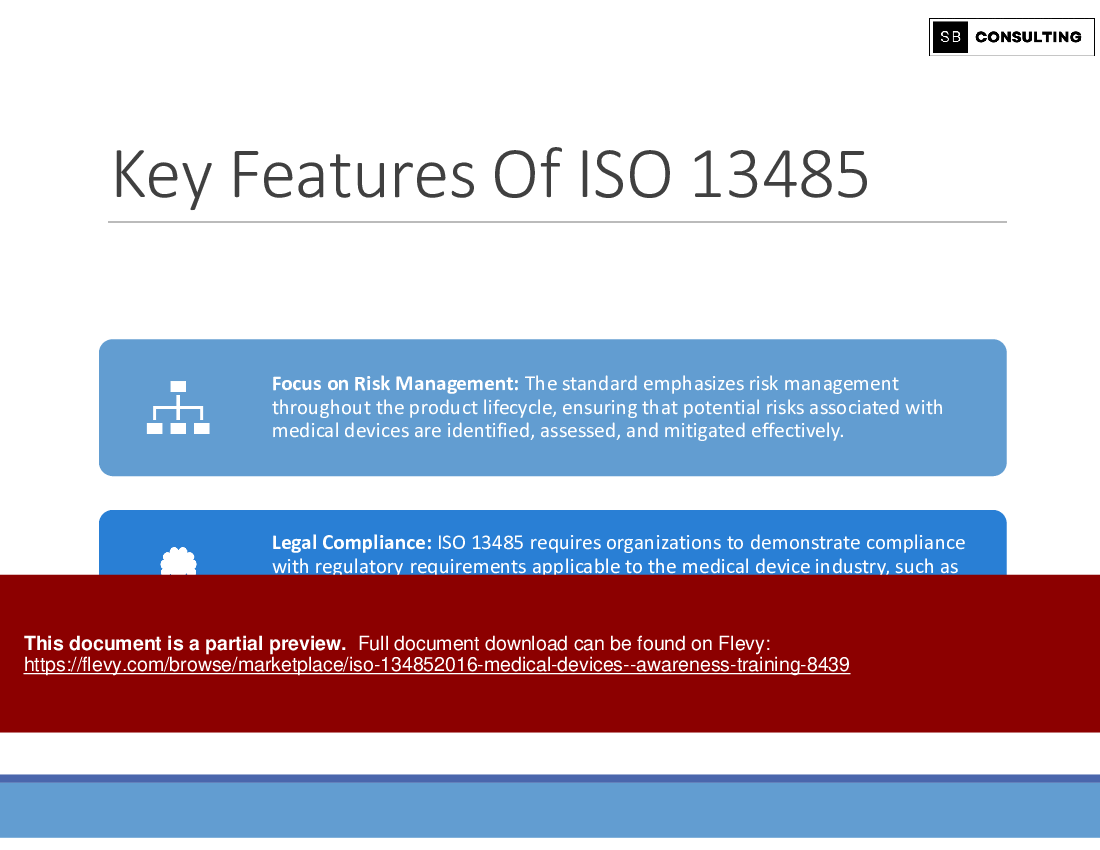
• Key Features of ISO 13485:2016
• Challenges of ISO 13485:2016
• Applications of ISO 13485:2016
• ISO 13485:2016 Scope
• PDCA (Plan-Do-Check-Act)
• ISO 13485:2016 Requirements
• ISO 13485:2016 Management Responsibilities
• Contamination Control
• Calibration and Verification
• ISO 13485:2016 Audit and Certification
• ISO 13485:2016 Audit Components
• ISO 13485:2016 Implementation
• ISO 13485:2016 Audit Checklists
• ISO 13485:2016 Case Study
LEARNING OBJECTIVES
Learning Objective 1: Understanding ISO and Standards
Embark on your learning journey by grasping the fundamental concepts of ISO and standards. Explore the significance of international standards in ensuring product quality, safety, and regulatory compliance within the medical device industry.
Learning Objective 2: Defining Medical Devices and Resource Management
Delve into the definition of medical devices and gain insights into effective resource management strategies. Understand how resource optimization plays a pivotal role in enhancing product quality, minimizing risks, and ensuring regulatory adherence.
Learning Objective 3: Exploring ISO 13485:2016 – An Overview
Unravel the essence of ISO 13485:2016 as the cornerstone of medical device quality management. Discover its evolution, principles, and relevance in fostering a culture of continual improvement and regulatory compliance.
Learning Objective 4: Unveiling the Benefits of ISO 13485:2016
Unlock the manifold benefits that ISO 13485:2016 offers to organizations operating in the medical device sector. From enhancing customer satisfaction to facilitating market access, explore how adherence to this standard can drive business success.
Learning Objective 5: Key Features and Challenges of ISO 13485:2016
Navigate through the key features and potential challenges associated with ISO 13485:2016 implementation. Gain insights into risk management, process documentation, and regulatory requirements while anticipating and overcoming common hurdles.
Learning Objective 6: Applications and Scope of ISO 13485:2016
Discover the diverse applications and expansive scope of ISO 13485:2016 across the medical device supply chain. From design and development to manufacturing and distribution, explore how this standard fosters quality at every stage.
Learning Objective 7: PDCA Cycle and ISO 13485:2016 Requirements
Master the Plan-Do-Check-Act (PDCA) cycle and delve into the specific requirements outlined in ISO 13485:2016. Acquire practical insights into establishing robust quality management systems that align with regulatory expectations.
Learning Objective 8: Management Responsibilities and Contamination Control
Understand the critical role of management in driving organizational compliance and fostering a culture of quality. Explore contamination control measures to mitigate risks and ensure product safety and efficacy.
Learning Objective 9: Calibration, Verification, and Audit
Navigate through the intricacies of calibration, verification, and audit processes mandated by ISO 13485:2016. Equip yourself with the knowledge to conduct internal audits, assess conformity, and prepare for external certification audits effectively.
Learning Objective 10: Implementation Strategies and Audit Checklists
Gain practical insights into effective implementation strategies tailored to your organization's unique needs. Access comprehensive audit checklists to streamline compliance efforts and facilitate successful ISO 13485:2016 certification.
Learning Objective 11: Case Study Analysis
Embark on a journey through real-world case studies highlighting successful ISO 13485:2016 implementations. Learn from industry best practices, challenges faced, and strategies employed to achieve compliance and drive business excellence.
Equip yourself with the knowledge, tools, and insights needed to excel in medical device quality management. Join us on this transformative learning experience with our ISO 13485 Awareness Training Toolkit and unlock the pathway to sustainable success in the dynamic healthcare landscape.
Key Words:
Strategy & Transformation, Growth Strategy, Strategic Planning, Strategy Frameworks, Innovation Management, Pricing Strategy, Core Competencies, Strategy Development, Business Transformation, Marketing Plan Development, Product Strategy, Breakout Strategy, Competitive Advantage, Mission, Vision, Values, Strategy Deployment & Execution, Innovation, Vision Statement, Core Competencies Analysis, Corporate Strategy, Product Launch Strategy, BMI, Blue Ocean Strategy, Breakthrough Strategy, Business Model Innovation, Business Strategy Example, Corporate Transformation, Critical Success Factors, Customer Segmentation, Customer Value Proposition, Distinctive Capabilities, Enterprise Performance Management, KPI, Key Performance Indicators, Market Analysis, Market Entry Example, Market Entry Plan, Market Intelligence, Market Research, Market Segmentation, Market Sizing, Marketing, Michael Porter's Value Chain, Organizational Transformation, Performance Management, Performance Measurement, Platform Strategy, Product Go-to-Market Strategy, Reorganization, Restructuring, SWOT, SWOT Analysis, Service 4.0, Service Strategy, Service Transformation, Strategic Analysis, Strategic Plan Example, Strategy Deployment, Strategy Execution, Strategy Frameworks Compilation, Strategy Methodologies, Strategy Report Example, Value Chain, Value Chain Analysis, Value Innovation, Value Proposition, Vision Statement, Corporate Strategy, Business Development
NOTE:Our digital products are sold on an "as is" basis, making returns and refunds unavailable post-download. Please preview and inquire before purchasing. Please contact us before purchasing if you have any questions! This policy aligns with the standard Flevy Terms of Usage.
Got a question about the product? Email us at support@flevy.com or ask the author directly by using the "Ask the Author a Question" form. If you cannot view the preview above this document description, go here to view the large preview instead.
Source: Best Practices in Healthcare, ISO 13485 PowerPoint Slides: ISO 13485:2016 Medical Devices - Awareness Training PowerPoint (PPTX) Presentation Slide Deck, SB Consulting